UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
Form
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For
the fiscal year ended:
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission
File Number
(Exact name of registrant as specified in its charter)
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) | |
| (Address of principal executive offices) | (Zip Code) |
| ( |
| (Registrant’s telephone number, including area code) |
SECURITIES REGISTERED PURSUANT TO SECTION 12(b) OF THE ACT:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| The Stock Market | ||||
| OTC Pink Open Market |
SECURITIES REGISTERED PURSUANT TO SECTION 12(g) OF THE ACT: NONE
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate
by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Exchange Act. Yes ☐
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2)
has been subject to such filing requirements for the past 90 days.
Indicate
by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule
405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant
was required to submit such files).
Indicate by check mark whether registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company, or an emerging growth company. See definitions of “large accelerated filer”, “accelerated filer”, “smaller reporting company”, and “emerging growth company” in Rule 12b-2 of the Exchange Act:
| Large accelerated filer ☐ | Accelerated filer ☐ |
| Smaller
reporting company | |
| Emerging
growth company |
If an emerging growth company, indicate by checkmark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to section 13(a) of the Exchange Act ☐
Indicate
by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness
of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered
public accounting firm that prepared or issued its audit report.
If
securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant
included in the fi ling reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No
The
aggregate market value of the common stock held by non-affiliates of the registrant on June 30, 2023, the last business day of the registrant’s
most recently completed second fiscal quarter, was $
Registrant had shares of common stock outstanding as of March 8, 2024.
DOCUMENTS INCORPORATED BY REFERENCE
ENSYSCE BIOSCIENCES, INC.
INDEX
| 2 |
Risk Factor Summary
Below is a summary of material factors that make an investment in our securities speculative or risky. Importantly, this summary does not address all of the risks and uncertainties that we face. Additional discussion of the risks and uncertainties in this summary, as well as other risks and uncertainties that we face, can be found under the section titled “Risk Factors” beginning on page 34 of this Annual Report on Form 10-K. This summary is qualified in its entirety by that more complete discussion of such risks and uncertainties. If we are unable to adequately address these and other risks we face, our business, results of operations, financial condition and prospects may be harmed. You should consider carefully the risks and uncertainties described under the section titled “Risk Factors” as part of your evaluation of an investment in our securities:
| ● | We are a clinical-stage pharmaceutical company with a limited operating history. We have incurred significant financial losses since our inception and anticipate that we will continue to incur significant financial losses for the foreseeable future. | |
| ● | We must obtain regulatory approval and fulfill numerous other requirements to be successful. | |
| ● | We require substantial additional funding. | |
| ● | There may be no proceeds under the GEM Agreement or proceeds may be less than anticipated. | |
| ● | We depend heavily on the success of PF614 and PF614-MPAR product candidates, which are currently in clinical trials, and which may not be successful. | |
| ● | Due to the significant resources required for the development of our product pipeline, and depending on our ability to access capital, we must prioritize the development of certain product candidates over others. | |
| ● | If we fail to discover, develop and commercialize other product candidates, we may be unable to grow our business. | |
| ● | If we do not achieve our projected development and commercialization goals within the timeframes we expect, the development and commercialization of our product candidates may be delayed. | |
| ● | Competitive products may reduce or eliminate potential commercial opportunity for our product candidates. | |
| ● | Our business could be harmed if we lose the services of our key personnel or if we are unable to hire additional highly qualified employees. | |
| ● | Our employees or others on which our business depends may engage in misconduct or other improper activities. | |
| ● | Government grant awards may not be available to us in the future. | |
| ● | We currently rely on, and expect to rely on in the future, third parties to conduct our clinical trials. | |
| ● | We expect to be completely dependent on third parties to manufacture our product candidates. | |
| ● | We must develop our sales, marketing, and distribution capability on our own or through collaborations. | |
| ● | The regulatory approval processes are lengthy, time-consuming, and inherently unpredictable. | |
| ● | Regulatory authorities may disagree with our regulatory plan for our product candidates. | |
| ● | Interim and preliminary data from our clinical trials may change. | |
| ● | We may incur unexpected costs or experience delays in completing, or ultimately be unable to complete, the preclinical and clinical studies necessary. | |
| ● | Even if we complete the necessary preclinical studies and clinical trials, the marketing approval process is expensive, time-consuming, and uncertain. | |
| ● | We may encounter difficulties enrolling patients in our clinical trials. |
| 3 |
| ● | Fast track designation by the FDA for PF614, for management of moderate to severe chronic pain when a continuous, around-the-clock analgesic is needed for an extended period of time, may not lead to a faster development or regulatory review or approval process and does not assure FDA approval. | |
| ● | If the FDA does not conclude that certain of our product candidates satisfy the requirements for the Section 505(b)(2) regulatory approval pathway, or if such requirements are not as we expect, the approval pathway will likely take significantly longer, cost significantly more and entail significantly greater complications and risks than anticipated, and in either case may not be successful. |
| ● | Changes in methods of product candidate manufacturing or formulation may result in additional costs or delay. | |
| ● | Our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following regulatory approval, if obtained. | |
| ● | Even if any of our product candidates receives regulatory approval, we may fail to achieve the degree of market acceptance necessary for commercial success. | |
| ● | We are subject to potential product liability lawsuits against us or any of our future collaborators. | |
| ● | Oxycodone is a Schedule II controlled substance under the federal CSA, and we must comply with the CSA or its state equivalents. | |
| ● | If we are unable to obtain and maintain patent protection for our products candidates, or if the scope of the patent protection obtained is not sufficiently broad, our competitors could develop and commercialize product candidates that are similar or identical to our product candidates. | |
| ● | We may face litigation from third parties claiming that our products or business infringe, misappropriate, or otherwise violate their intellectual property rights, or seeking to challenge the validity of our patents. | |
| ● | We may become involved in lawsuits to protect or enforce our patents or other intellectual property. | |
| ● | The expiration or loss of patent protection may adversely affect our future revenues and operating earnings. | |
| ● | We may not be able to obtain protection under the Hatch-Waxman Amendments by extending the patent term. | |
| ● | We may not be able to protect our intellectual property rights throughout the world. | |
| ● | We may be subject to claims that we infringed, misappropriated or otherwise violated the intellectual property of a third party, or claiming ownership of what we regard as our own intellectual property. | |
| ● | Our intellectual property agreements with third parties may be subject to disagreements. | |
| ● | Intellectual property rights do not necessarily address all potential threats to our business. | |
| ● | Cyber-attacks or other failures in our telecommunications or information technology systems, or those of third parties could result in information theft, data corruption and significant disruption of our business. | |
| ● | Raising additional capital in the public or private equity markets at prices per share below the current market price of our common stock could cause dilution to our stockholders, adversely affect the market price of our common stock, restrict our operations or require us to relinquish rights to our technologies or product candidates. | |
| ● | Our internal controls over financial reporting currently do not meet all of the standards contemplated by Section 404 of Sarbanes-Oxley Act. | |
| ● | Nasdaq may delist our common stock and/or our Public Warrants may not continue to trade on the OTC Pink Open Market. |
| 4 |
GLOSSARY
| Definitions: | ||
| 2013 Framework | Financial reporting criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework (2013) | |
| 2021 Notes | The senior secured convertible promissory notes in the aggregate original principal amount of $15.9 million, sold in two closings on September 24, 2021 and November 5, 2021, respectively, pursuant to the Securities Purchase Agreement entered into on September 24, 2021 | |
| 2021 Omnibus Incentive Plan | Ensysce Biosciences, Inc. Amended and Restated 2021 Omnibus Incentive Plan | |
| 2022 Notes | The senior secured convertible promissory notes in the aggregate original principal amount of $8.5 million, sold in two closings on June 30, 2022 and August 8, 2022, respectively, pursuant to the Securities Purchase Agreement entered into on June 30, 2022 | |
| 2023 Notes | The senior secured convertible promissory notes in the aggregate original principal amount of $1.8 million, sold in two closings on October 25, 2023 and November 28, 2023, respectively, pursuant to the Securities Purchase Agreement entered into on October 23, 2023 | |
| 2022 December Offering | The Company’s December 2022 registered direct offering of common stock (including pre-funded warrants in lieu thereof). The Offering closed on December 9, 2022 for aggregate consideration of $4.1 million | |
| 2023 February Offering | The Company’s February 2023 registered direct offering of common stock and private placement warrants for aggregate consideration of $3.0 million | |
| 2023 May Offering | The Company’s May 2023 registered direct offering of common stock (including pre-funded warrants in lieu thereof) for aggregate consideration of $7.0 million | |
| ADFs | Abuse deterrent formulations | |
| ADHD | Attention deficit hyperactivity disorder | |
| Aggregate Limit | Up to $60 million of gross proceeds with respect to the GEM Agreement | |
| ANDA | Abbreviated New Drug Application | |
| API | Active pharmaceutical ingredient | |
| AUC | Area under the concentration time curve | |
| Board | Board of directors of Ensysce, or a committee thereof, as applicable | |
| Business Combination | The definitive merger agreement among LACQ, Merger Sub and Former Ensysce, dated January 31, 2021, providing for, among other things, and subject to terms and conditions therein, the business combination between LACQ and Former Ensysce pursuant to the merger of Merger Sub with and into Former Ensysce, with Former Ensysce continuing as the surviving entity and as a wholly-owned subsidiary of LACQ | |
| CARA | Comprehensive Addiction and Recovery Act | |
| CDC | Center for Disease Control | |
| CDER | Center for Drug Evaluation and Research | |
| cGMP | Current Good Manufacturing Practice | |
| Cmax | Maximum plasma concentration | |
| CMC | Chemistry, manufacturing, and controls | |
| CMOs | Contract manufacturing organizations | |
| CNS | Central nervous system | |
| Company | Ensysce Biosciences, Inc. and its consolidated subsidiaries | |
| COVID-19 | Novel coronavirus disease | |
| Covistat | A subsidiary renamed EBIR, Inc. | |
| CROs | Contract research organizations | |
| CSA | Controlled Substances Act | |
| CSOS | Controlled Substance Ordering System | |
| DEA | United States Drug Enforcement Agency | |
| Draw Down Limit | 400% of the average daily trading volume for the 30 trading days immediately preceding the date the Company delivers the draw down notice with respect to the GEM Agreement |
| 5 |
| DSCSA | Title II of the Federal Drug Quality and Security Act of 2013, known as the Drug Supply Chain Security Act | |
| EB | Ensysce Biosciences, Inc. prior to its merger with Signature Acquisition Corp. pursuant to the EB-ST Agreement. | |
| EBIR | Previously known as Covistat, Inc., EBIR, Inc. is a clinical stage pharmaceutical company that is developing a compound utilized in the Company’s overdose protection program for the treatment of COVID-19 and 79.2%-owned subsidiary of the Company | |
| EB-ST Agreement | Agreement and Plan of Merger, dated as of December 28, 2015, by and among Signature, SAQ, and EB | |
| EMA | European Medicines Agency | |
| Ensysce | Ensysce Biosciences Inc. | |
| EPO | European Patent Office | |
| ETASU | Elements to assure a products safe use | |
| Exchange Act | Securities Exchange Act of 1934, as amended | |
| FDA | United States Food and Drug Administration | |
| FDC Act | Federal Food, Drug and Cosmetic Act, as amended | |
| Former Ensysce | Ensysce Biosciences, Inc., a Delaware corporation, prior to the consummation of the merger with and into Merger Sub | |
| GAAP | Generally Accepted Accounting Principles in the United States of America | |
| GCP | Good Clinical Practices | |
| GEM Agreement | Share Purchase Agreement between the Company, GEM Global, and GYBL, dated as of December 29, 2020, including a Registration Rights Agreement between the same parties and dated as of the same date | |
| GEM Global | GEM Global Yield LLC SCS | |
| GEM Warrants | 4,608 shares of common stock that may be issued upon the exercise of warrants issued to GYBL under the terms of the GEM Agreement at an exercise price of $1.5675 per share | |
| GMP | Good Manufacturing Practices | |
| GYBL | GEM Yield Bahamas Limited | |
| Hatch-Waxman Act or Hatch-Waxman Amendments | Drug Price Competition and Patent Term Restoration Act of 1984 | |
| HHS | United States Department of Health and Human Services | |
| IMPDs | Investigational Medicinal Product Dossiers | |
| IND | Investigational New Drug | |
| Investor Notes | The 2021 Notes, 2022 Notes and 2023 Notes, collectively. | |
| IRB | Institutional Review Board | |
| JOBS Act | Jumpstart Our Business Startups Act of 2012 | |
| LACQ | Leisure Acquisition Corp., a Delaware Corporation | |
| LACQ Warrants | Warrants that relate to the Business Combination or were issued prior to it and are exercisable for 21,993 shares of our common stock at a weighted average exercise price of $2,725.90 per share | |
| MAD | Multi-Ascending Dose | |
| Merger | The merger of Merger Sub with and into Former Ensysce, with Former Ensysce continuing as the surviving entity and a wholly owned subsidiary of LACQ, which changed its name to Ensysce Biosciences, Inc. following consummation of the Merger. | |
| Merger Agreement | Agreement and Plan of Merger, dated as of January 31, 2021, by and among LACQ, Merger Sub and Former Ensysce, providing for, among other things, and subject to the terms and conditions therein, a business combination between Former Ensysce and LACQ pursuant to the proposed merger of Merger Sub with and into Former Ensysce, with Former Ensysce surviving the transaction as a wholly-owned subsidiary of LACQ, which changed its name to Ensysce Biosciences, Inc. following consummation of the Merger | |
| Merger Sub | EB Merger Sub, Inc., a Delaware corporation, a wholly-owned subsidiary of LACQ prior to the consummation of the Merger |
| 6 |
| MPAR Grant | Research and development grant related to the development of its MPAR® overdose prevention technology awarded to the Company by NIH through NIDA in September 2018 | |
| Nasdaq | The Nasdaq Stock Market LLC | |
| NCE | New Chemical Entity | |
| NDA | New Drug Application | |
| NEO | Named Executive Officer | |
| NIDA | National Institute of Drug Abuse | |
| NIH | National Institutes of Health | |
| NME | New molecular entity | |
| Orange Book | FDA’s publication Approved Drug Products with Therapeutic Equivalence Evaluations | |
| OUD Grant | Research and development grant related to the development of its TAAP/MPAR® abuse deterrent technology for Opioid Use Disorder awarded to the Company by NIH/NIDA in September 2019 | |
| PCT | Patent Cooperation Treaty | |
| PDMA | U.S. Prescription Drug Marketing Act | |
| PK | Pharmacokinetics | |
| Prior Warrants | Warrants issued pursuant to the Securities Purchase Agreement. The Prior Warrants issued in (i) 2021 are exercisable for an aggregate of 4,518 shares of our common stock at an exercise price of $3.64 per share and in (ii) 2022 are exercisable for an aggregate of 38,900 shares of our common stock at an exercise price of $3.64 per share | |
| PTA | Patent Term Adjustment | |
| PTE | Patent Term Extension | |
| Public Warrants | The redeemable warrants issued by us and sold as part of the units in the LACQ IPO (whether they were purchased in the LACQ IPO or thereafter in the open market). The Public Warrants are exercisable for an aggregate of approximately 41,666 shares of our common stock at an exercise price of $2,760.00 per share | |
| R&D | Research and Development | |
| Recro | Recro Gainesville LLC | |
| Recro Agreement | Manufacturing Agreement, dated September 19, 2019, by and between Recro Gainesville LLC and the Company | |
| REMS | Risk evaluation and mitigation strategy | |
| Resale Registration Statement | Ensysce’s Resale Registration Statement filed on August 9, 2021 | |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 | |
| SAQ | Signature Acquisition Corp., a wholly-owned subsidiary of Signature | |
| SEC | U.S. Securities and Exchange Commission | |
| Securities Act | Securities Act of 1933, as amended | |
| Securities Purchase Agreement | The Securities Purchase Agreement, dated as of September 24, 2021, June 30, 2022, or October 23, 2023 as the context dictates, by and between Ensysce and the institutional investors party thereto | |
| Signature | Signature Therapeutics Inc. | |
| SPA | A Securities Purchase Agreement, dated as of September 24, 2021, June 30, 2022, or October 23, 2023 as the context dictates, by and between Ensysce and the institutional investors party thereto | |
| SUPPORT Act | Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act | |
| TAAP | Trypsin Activated Abuse Protection | |
| TEAEs | Treatment-emergent adverse events | |
| USPTO | United States Patent and Trademark Office |
| 7 |
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K includes statements that express our opinions, expectations, beliefs, plans, objectives, assumptions, or projections regarding future events or future results and therefore are, or may be deemed to be, “forward-looking statements.” These forward-looking statements can generally be identified by the use of forward-looking terminology, including the terms “believes,” “estimates,” “anticipates,” “expects,” “seeks,” “projects,” “intends,” “plans,” “may,” “will,” or “should” or, in each case, their negative or other variations or comparable terminology. These forward-looking statements include all matters that are not historical facts. They appear in a number of places throughout this Annual Report on Form 10-K and include statements regarding our intentions, beliefs or current expectations concerning, among other things, results of operations, financial condition, liquidity, prospects, growth, strategies and the markets in which we operate. Such forward-looking statements are based on available current market material and management’s expectations, beliefs and forecasts concerning future events impacting our company. Factors that may impact such forward-looking statements include:
| ● | our estimates regarding expenses, revenue, capital requirements and timing and availability of and the need for additional financing will almost certainly not match actual amounts and timing; | |
| ● | our ability to continue as a going concern for the next twelve months; | |
| ● | the risk that our lead product candidate PF614 and PF614-MPAR may not be successful in limiting or impeding abuse, overdose, or misuse or providing additional safety upon commercialization; | |
| ● | reliance by us on third-party contract research organizations, or CROs, for our research and development activities and clinical trials; | |
| ● | the need for substantial additional funding to complete the development and commercialization of our product candidates; | |
| ● | the risk that our clinical trials may fail to replicate positive results from earlier preclinical studies or clinical trials conducted by us or third parties; | |
| ● | the risk that the potential product candidates that we develop may not progress through clinical development or receive required regulatory approvals within expected timelines or at all; | |
| ● | the risk that clinical trials may not confirm any safety, potency, or other product characteristics described or assumed in this Annual Report on Form 10-K; | |
| ● | the risk that we will be unable to successfully market or gain market acceptance of our product candidates; | |
| ● | the risk that our product candidates may not be beneficial to patients or successfully commercialized; | |
| ● | the risk that we have overestimated the size of the target market, patients’ willingness to try new therapies, and the willingness of physicians to prescribe these therapies; | |
| ● | effects of competition; |
| ● | the risk that third parties on which we depend for laboratory, clinical development, manufacturing, and other critical services will fail to perform satisfactorily; | |
| ● | the risk that our business, operations, clinical development plans and timelines, and supply chain could be adversely affected by the effects of health epidemics; | |
| ● | the risk that we will be unable to obtain and maintain sufficient intellectual property protection for its investigational products or will infringe the intellectual property protection of others; |
| ● | the loss of key members of our management team; | |
| ● | changes in our regulatory environment; | |
| ● | the ability to attract and retain key scientific, medical, commercial, or management personnel; | |
| ● | changes in our industry; | |
| ● | our ability to remediate any material weaknesses or establish and maintain effective internal controls over financial reporting; | |
| ● | the risk that our common stock will be delisted from Nasdaq; | |
| ● | the risk that we may not be able to regain or maintain compliance with applicable listing standards of Nasdaq; | |
| ● | potential litigation associated with the Business Combination Transactions; | |
| ● | other factors disclosed in this Annual Report on Form 10-K; and | |
| ● | other factors beyond our control. |
The forward-looking statements contained in this Annual Report on Form 10-K are based on our current expectations and beliefs concerning future developments and their potential effects on our company. There can be no assurance that future developments affecting us will be those that we have anticipated. These forward-looking statements involve a number of risks, uncertainties (some of which are beyond our control) or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to, those factors described under the heading “Risk Factors”. Should one or more of these risks or uncertainties materialize, or should any of the assumptions prove incorrect, actual results may vary in material respects from those projected in these forward-looking statements. We will not undertake any obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as may be required under applicable securities laws.
| 8 |
References in this Annual Report on Form 10-K to “we,” “our,” “us,” the “Company” or “Ensysce” generally refer to Ensysce and its consolidated subsidiaries.
PART I
Item 1. Business
Corporate Information
We were originally incorporated in the State of Delaware in April 2003 as PharmacoFore, Inc. and, in January 2012, we changed our name from PharmacoFore, Inc. to Signature Therapeutics Inc. (“Signature”). On December 28, 2015, Signature, Signature Acquisition Corp., a wholly-owned subsidiary of Signature (“SAQ”), and Ensysce Biosciences, Inc. (“EB”) entered into an Agreement and Plan of Merger (“EB-ST Agreement”). Pursuant to the EB-ST Agreement, SAQ merged with and into EB with EB surviving the merger as a wholly-owned subsidiary of Signature. As part of the transaction, Signature changed its name to “Ensysce Biosciences, Inc.” (“Former Ensysce”) and changed EB’s name to EBI Operating Inc. On January 31, 2021, LACQ, Former Ensysce, and Merger Sub entered into the Merger Agreement. On June 30, 2021, pursuant to the Merger Agreement, Merger Sub merged with and into Former Ensysce, with Former Ensysce surviving the transaction as a wholly-owned subsidiary of LACQ. As part of the transaction, LACQ changed its name to “Ensysce Biosciences, Inc.” and Former Ensysce changed its name to EBI OpCo, Inc.
The mailing address of our principal executive office is 7946 Ivanhoe Avenue, Suite 201, La Jolla, California 92037. Our corporate telephone number is (858) 263-4196. Our website address is www.ensysce.com. Information contained on our website, or connected thereto, does not constitute part of, and is not incorporated by reference into, this Annual Report on Form 10-K.
Channels for Disclosure of Information
Investors, the media, and others should note that we announce material information to the public through filings with the SEC, the investor relations page on our website, blog posts on our website, press releases, public conference calls, webcasts, and our twitter feed (@EnsysceBio).
The information disclosed by the foregoing channels could be deemed to be material information. As such, we encourage investors, the media, and others to follow the channels listed above and to review the information disclosed through such channels.
Any updates to the list of disclosure channels through which we will announce information will be posted on the investor relations page on our website.
Business Overview
We are a clinical stage pharmaceutical company seeking to develop innovative solutions for severe pain relief while reducing the potential for opioid misuse, abuse, and overdose.
We are currently developing product candidates designed to improve the safety of prescription drugs. Our primary focus has been on opioid pain products and opioid use disorder products. Prescription opioid abuse presents major burdens to society, resulting in significant costs, illnesses, and deaths, many of which we believe could be prevented through the use of our proprietary technologies. We believe the intertwined issues of (1) the widespread abuse of prescription opioids and (2) the resultant reluctance of many prescribers to write prescriptions for opioid analgesics have resulted in the persistent under-treatment of patients with moderate-to-severe pain. Our platforms utilize a novel molecular delivery technology designed to deter prescription opioid abuse at the molecular level.
Our current development pipeline includes two new drug platforms - an abuse-resistant opioid prodrug technology – the Trypsin Activated Abuse Protection, or the TAAP platform, and an over-dose protection opioid prodrug technology - the Multi-Pill Abuse Resistant, or the MPAR® platform. The TAAP platform is designed to seek to improve the care of patients with chronic pain while reducing the human and economic costs associated with prescription opioid drug abuse. The MPAR® platform when combined with our TAAP prodrugs is designed not only to seek to prevent abuse of prescription drugs but also to reduce overdose occurrences. Each prodrug is intended to be able to be combined with our MPAR® technology for overdose protection. Additionally, nafamostat di-mesylate (“nafamostat”), which is an ingredient in our overdose protection combination products, is also being developed for the intended purpose of treating infection and pulmonary lung diseases.
| 9 |
The technology under the TAAP platform when applied to opioid drugs is designed to release clinically effective opioid drugs only when exposed to specific physiological conditions (i.e., when the drug is ingested and exposed to the digestive enzyme trypsin). Our lead product candidate, PF614, is a TAAP oxycodone prodrug that is a biologically inactive compound which can be metabolized in the body to produce a drug with demonstrable features aimed at resisting both oral and non-oral modes of prescription drug abuse. This approach differs from current formulation-based strategies such as OxyContin OP which uses Intac® Technology (crush-resistant polymers) and Extampza®ER which uses DETERx™ (insoluble fatty acid salts in polymers), in a number of ways. First, the TAAP technology seeks to remove the ability of a user to abuse PF614 intravenously or intranasally based on preclinical studies that show PF614 does not readily convert into oxycodone in the blood stream and trypsin is not present in the nasal passage, and, accordingly, PF614 would not convert to oxycodone in the nose. Furthermore, the chemically modified and abuse-resistance TAAP opioid drug is unaffected by simple physical manipulations designed to extract abusable amounts of opioid, such as through kitchen chemistry. Our portfolio of TAAP product candidates is based on a differentiated understanding of chemical reactivity and metabolism, as well as the key pillars of our unique approach which focuses on: (1) enzyme mediated metabolic activation localized in the gastrointestinal track; (2) rearrangement chemistry to achieve pharmacokinetic release of active drug products; and (3) preclinical and clinical data that set forth the metabolic and chemical activation profile for each of our clinical candidates.
Utilizing this approach, we filed an Investigational New Drug application, or IND (116794), and commenced a Phase 1 clinical trial for PF614, which was completed in February 2018. The clinical data from the Phase 1 trial demonstrated that oxycodone released from PF614 as chemically-designed, and that it was absorbed following oral administration of the TAAP PF614, resulting in blood levels that matched the same release profile as the extended release oxycodone product, OxyContin OP. A second multi-ascending dose study with a bioequivalent arm was completed in July 2022 and a nasal human abuse potential (HAP) study was completed in October 2022. A second oral HAP study was completed in March 2023. Most recently, a study to evaluate efficacy was completed in December 2023.
The MPAR® technology is designed to limit the bioavailability of active opioid following co-ingestion of multiple doses, whether inadvertent or intentional, through a combination of a TAAP prodrug with nafamostat. Nafamostat is a small molecule that clinical studies have shown to have a steep dose response curve and to be a highly potent trypsin inhibitor. When combined with our TAAP prodrugs, our MPAR® technology is designed not to affect metabolism and the release of the active pharmaceutical ingredient. However, if the MPAR® combination product is taken in larger quantities than intended, the excess nafamostat is designed to inhibit trypsin, thereby preventing metabolic activation and averting a drug overdose. We believe the potential benefits to society of an opioid that resists both oral and parenteral abuse are considerable. A Phase 1 study to explore the combination of PF614 and nafamostat, PF614-MPAR-101 was initiated in December of 2021, and early data from the study reported in May 2022 demonstrated the combination product showed overdose protection, with a reduction in the release of oxycodone over that of PF614 delivered alone. A second part of this trial to confirm overdose protection from PF614-MPAR 25 mg was completed in 2023 and data reported in May 2023. PF614-MPAR was granted Breakthrough Therapy designation by the FDA in January 2024.
Our pipeline has been developed over the course of 15 years of research and investment and includes three clinical-stage product candidates. While our principal focus and lead product candidates are geared towards combating abuse and overdose of opioid drugs, we have, over the years of research and development, discovered and recognized qualities and unique features of certain product candidates that may be useful in addressing other treatments. For example, we discovered the ability of nafamostat in inhibiting the action of enzymes associated with the COVID-19 infection, and, as such, have devoted efforts to develop an oral and inhalation drug product of nafamostat, for use against coronaviral infections and other pulmonary diseases such as cystic fibrosis.
| 10 |
PF614
PF614 is our lead TAAP prodrug candidate under development for the treatment of acute or chronic pain. PF614 is a delayed release TAAP prodrug designed to release oxycodone under certain specific physiological circumstances when taken orally. PF164 was evaluated for safety and pharmacokinetic release of oxycodone in a Phase 1 single ascending dose clinical trial in 64 healthy subjects. The trial showed that PF614 was well tolerated with no serious adverse events. The study also showed pharmacokinetics had a maximum blood concentration of oxycodone at 4 to 6 hours after swallowing PF614, demonstrating its delayed release profile. A second Phase 1b multi-ascending dose study (MAD) was initiated in 2021 to evaluate PF614 delivered to healthy subjects twice daily for 4.5 days. This study evaluated both safety and PK, with a second part to evaluate the bioequivalence (BE) of PF614 versus OxyContin. Final data from this trial was reported in July 2022. The MAD study demonstrated both the safety of PF614 showing it was well tolerated at doses up to 200 mg, which was comparable to 80 mg of OxyContin both delivered twice daily. The BE study arm followed the successful completion of the multi-ascending twice-daily dosing study of PF614 and compared the release of oxycodone from PF614 versus OxyContin® administered to subjects in both fasted and fed states. It was concluded that 100 mg PF614 was bioequivalent to 40 mg OxyContin under both fasted and fed conditions. This data is critical to understand future prescribing criteria for PF614 as an agent bioequivalent to OxyContin and therefore may be developed through the 505(b)(2) regulatory path as defined by the FDA. The intranasal (IN) and oral human abuse potential of PF614 was assessed in two different studies. In study 1, PF614-103, we evaluated the abuse potential of PF614 100 mg relative to crushed oxycodone immediate-release (IR) tablets 40 mg (equivalent opioid doses) and placebo following intranasal administration. In study 2, PF614-104, we evaluated the oral abuse potential of intact PF614 at 3 different dose levels 50, 100 and 200 mg to IR oxycodone 40 mg and placebo. The purpose of this study is to assess the PK and human oral abuse potential of PF614. An efficacy study, PF614-201 was conducted to evaluate 50 and 100 mg PF614 in 16 healthy male subjects for the time of onset of pain relief and the ability of PF614 to relieve pain. The data will be used to design our Phase 3 clinical trials. We believe PF614 has the potential to provide a safer alternative to the abuse deterrent formulated opioid products that are currently commercially available.
PF614-MPAR
PF614-MPAR, a combination product of PF614 and nafamostat has been designed to limit abuse potential by providing resistance to use through injection or inhalation and to provide overdose protection against excessive oral ingestion. Our IND application (150966) for PF614-MPAR received FDA allowance on April 27, 2021 following the release of a Full Clinical Hold from January 8, 2021. We addressed deficiencies from the initial IND submission, amended the protocol and submitted a response to the clinical hold letter on March 29, 2021. We initiated a Phase 1 clinical trial, PF614-MPAR-101, to evaluate safety and PK in healthy subjects in December 2021. Initial data from this trial was reported in May of 2022. The PF614-MPAR-101 overdose protection study examined PF614 administered orally alone or in combination with the trypsin inhibitor nafamostat (MPAR®) to healthy volunteers. The initial data demonstrated the overdose protection of our MPAR® combination product, with reduced release of oxycodone from PF614 in a simulated overdose situation. It also demonstrated the PF614 in the systemic circulation (simulated injection) did not convert to oxycodone. We completed the clinical portion and reported data from Part A of this study in December 2022. Part B of the study to test the overdose protection of the selected PF614-MPAR 25 mg formulation by administering an escalating number of dose units to a group of healthy subjects completed enrollment in March 2023. The PK data from Part B successfully showed that PF614-MPAR® 25 mg administered at a prescribed dose of one or two dose units (capsules) provided oxycodone in an equivalent manner to PF614 without MPAR®. However, the simultaneous administration of 3 dose units or greater of PF614-MPAR 25 mg, resulted in reduced oxycodone in the circulation, as compared to the unprotected PF614. There was a highly significant difference between the oxycodone blood levels following delivery of PF614 200 mg versus 8 dose units of PF614-MPAR 25 mg (200 mg PF614 total) demonstrating the overdose protection produced by the MPAR® technology. This was reported in May of 2023. PF614-MPAR was granted Breakthrough Therapy designation by the FDA in January 2024.
MPAR is being tested clinically in partnership with Quotient Sciences, using its integrated Translational Pharmaceutics® platform to search for a PF614-MPAR formulation that allows conversion into oxycodone within the prescribed dose range but reduces conversion to oxycodone at higher than prescribed dose levels in an overdose scenario.
Nafamostat
Nafamostat is an enzyme inhibitor (protease inhibitor) used in our combination overdose protection technology, MPAR®. Due to its ability to inhibit the action of enzymes associated with the COVID-19 infection, we are also developing an oral drug product of nafamostat, for use against coronaviral infections and other pulmonary diseases such as cystic fibrosis. An IND was submitted (149877) for the evaluation of oral nafamostat in coronaviral infections. A Phase 1 trial to evaluate safety and PK was completed in 2021.
| 11 |
Market Opportunity
Drug Abuse and Drug Overdose
Opioid pain medications are essential for improving the care and outcomes of a majority of Americans who live with chronic pain. An NIH study, updated September 2018, reported that 25.3 million adults suffered from pain every day for the preceding three months and almost 40 million adults experience severe levels of pain, which is linked to worse health status. High impact chronic pain affects over 10 million Americans and is characterized by extended periods of suffering which impair life quality to a severe degree. Prescription opioids drugs, such as morphine, hydromorphone, hydrocodone, and oxycodone, have a long history of use for the management of severe and chronic pain. Prescriptions for opioid medications in 2021 totaled 153 million, with $4.2 billion in market size in the United States.
The CDC recently provided recommendations for clinicians who provide pain care, defining acute pain (duration less than 1 month), subacute pain (duration of 1–3 months), or chronic pain (duration of 3 months or more), not including sickle cell disease related pain management, cancer pain treatment, palliative care, and end-of life care. These guidelines are based on the indications, acute and chronic pain, that we intend to explore for our TAAP and MPAR® opioid products including PF614.
Opioids are offered in a variety of dosages including immediate-release tablets (or capsules), extended-release tablets (or capsules), patches, and other dose forms. Oxycodone is one of the most effective pain killers available today. This drug helps the patient to overcome pain and focus on his or her work. Opioids have a risk of dependence and, when used improperly, a common side effect of high doses of opioids like oxycodone can be euphoria, or a “high.” As a result of these side effects, opioids have become amongst the most misused or abused prescription drugs in the United States. Opioid abuse was declared a public-health emergency in 2017 when more than 91 people died each day from opioid-related overdoses. In 2021 the total number of opioid-related deaths rose to 109,600, with 45 people dying each day from a prescription opioid overdose.
The large increase in overall overdose deaths is now driven by use of synthetic opioids, in particular fentanyl, as prescription opioids have become harder to obtain. From 2017 to 2018 the prescription opioid-involved death rates decreased by 13.5% showing that attention to the problem had beneficial effect. However, 2.1 million people reported having opioid use disorder (“Opioid Use Disorder”) in 2019. Based on information from the CDC, the most common drugs involved in prescription opioid overdose deaths include Methadone, Oxycodone (such as OxyContin®), and Hydrocodone (such as Vicodin®). The CDC indicates that improving opioid prescribing, treatment of opioid use disorder, and prevention of opioid use disorder would help to improve the opioid crisis. Misuse or abuse of opioids is often done in one of the following manners:
| ● | Oral Excessive Tablet Abuse. Generally recognized as the most prevalent route of administration by abusers, an abuser orally ingests more tablets (or capsules) than is recommended for pain relief. | |
| ● | Nasal snorting. Crushed tablets are insufflated for absorption of the drug through the nasal tissues. | |
| ● | Injection. The opioid is physically or chemically removed from the dosage and injected into the vein using a syringe. | |
| ● | Oral Manipulated Tablet Abuse. Extended-release tablets or patches are crushed, chewed, or otherwise physically or chemically manipulated to defeat an extended-release mechanism and provide an immediate-release of the opioid for oral ingestion. | |
| ● | Poly-pharmacy. Opioids are sometimes used in conjunction with alcohol, methamphetamine, benzodiazepines or other drugs to enhance the euphoria. |
| 12 |
| ● | Overdose. Users may accidentally introduce excessive quantities of drugs in their systems or combine drugs that may heighten the chance of adverse effects of drugs. Some patients may over-ingest drugs accidentally or with the express intent of suicide. | |
| ● | Chronic or prolonged use. Chronic or prolonged use of opioids resulting in dependence is another form of misuse or abuse. |
Amphetamines like Adderall are manufactured in pill form and are intended for oral ingestion. As of Q4 2022, seventy-five percent of Adderall prescriptions are prescribed to the 10.5 million adults, age 22 or older, that are diagnosed with attention deficit hyperactivity disorder, or ADHD. ADHD is the most common neurodevelopment disorder in children. Five million adults misuse stimulant medication annually, by using alternative consumption methods to achieve a more intense high faster; snorting or injecting are most-common methods of abuse. Both of these methods involve crushing pills.
We believe that having prescription drug products available that have a reduced potential for abuse by crushing and injecting, snorting, and chewing could provide an even greater reduction of prescription opioid related deaths in the abuse of opioids or amphetamines.
Nafamostat
Nafamostat’s market opportunity is multifaceted. The oral form could be used alone or in combination with other antiviral drugs that target separate processes needed for virus product, such as RNA replication or viral protein processing. Nafamostat delivered orally was evaluated in a Phase 1 study for safety and tolerability with the anticipation of further evaluating an oral nafamostat drug product against COVID-19 in Phase 2 trials, however at this time the oral nafamostat program is not the company’s primary focus.
Our Technology Platform Solution
TAAP Prescription Drugs
The technology under the TAAP platform utilizes a novel technology designed to deter prescription drug abuse at the molecular level. The molecular delivery system is designed to release clinically effective drugs only when exposed to specific physiological conditions (i.e., when the drug is ingested and exposed to the digestive enzyme trypsin). Our TAAP prodrugs delivery system demonstrates a number of features aimed at resisting both oral and non-oral modes of abuse. This platform’s approach differs from current formulation-based strategies (abuse deterrent formulations, or ADFs) in a number of ways. First, the abuse-resistance provided by TAAP is designed to be unaffected by simple physical manipulations (e.g., crushing and extraction and/or chewing of the dose form provided to patients). We believe the potential benefits to society of applying TAAP to opioids and amphetamines providing medication that resists both oral and parenteral abuse are considerable.
MPAR® Prescription Drugs
MPAR® combination therapy, involves co-formulating TAAP prodrugs with a trypsin inhibitor, nafamostat, which, when administered at prescribed dose levels, are intended to have no effect on the conversion of the prodrug to the active ingredient thus allowing normal drug plasma exposure levels. However, if the drug were taken in greater than prescribed quantities, the trypsin inhibitor would also be present at higher levels, inhibiting the first step in the activation process, preventing the conversion of the prodrug to the active ingredient thus limiting the potential to an overdose from the medication.
Our Development Programs
We are currently developing product candidates designed to improve the safety and performance of prescription drugs. Our primary focus has been on opioid pain products and opioid use disorder products. Each prodrug is intended to be able to be combined with our MPAR® technology for overdose protection. Additionally, nafamostat, which is an ingredient in our overdose protection combination products, may be developed for infection and pulmonary lung diseases. Besides our clinical candidates, we have a product portfolio of other TAAP and MPAR® opioids and amphetamines that could potentially be developed to build on this pipeline.
| 13 |
Clinical agents
PF614
PF614 is a chemically modified, extended-release oxycodone-derivative which releases clinically effective oxycodone only when exposed trypsin in the gut (i.e., when the drug is ingested). This approach differs from formulation-based strategies which are currently commercially available, in a number of ways. First, the abuse-resistance provided by PF614 is retained even when dissolved in water and is designed to be unaffected by simple physical manipulations (e.g., extraction, chewing, and/or crushing). It also limits the bioavailability of active medication following co-ingestion of multiple doses.
Following ingestion, the release of oxycodone from PF614 proceeds via a two-step process comprised of (1) trypsin activation in the small intestine and (2) a subsequent intramolecular cyclization release reaction. This reaction releases oxycodone with concomitant formation of a cyclic urea metabolite. The time-course of oxycodone release from PF614 is a function of the kinetics of (i) the trypsin hydrolysis and (ii) the cyclization-release reaction. In the Phase 1 study of PF614, the time to maximal blood concentration of oxycodone (Tmax) was five to six hours for the release of oxycodone and this time cannot be modified by crushing, chewing, or physically manipulating the drug product. Oxycodone safety, metabolism, and pharmacokinetics have been well studied.
PF614-101 Phase 1 Clinical Trial
PF614 (IND 116796) has been evaluated in a Phase 1 clinical study for safety and pharmacokinetics of oxycodone release in 64 healthy subjects in seven different closing cohorts from November 2016 to January 2018. This study was conducted for us by PRA Health Sciences – Early Development Services Lenexa, Kansas, principal investigator, Daniel Dickerson, M.D., Ph.D. to evaluate the safety and pharmacokinetics of PF614, as well as the pharmacokinetics of oxycodone at doses sufficient to characterize the extent to which plasma oxycodone is produced and maintained following oral ingestion of PF614 and was compared to the oxycodone released from extended release oxycodone from OxyContin OP. Subjects were randomized to receive a single dose of PF614 (dose of 15, 25, 50, 100, and 200 mg with 6 subjects per dosing group) or OxyContin OP (dose of 10, 20, 50, and 80 mg with 2 subjects per dosing group). New subjects were recruited for each cohort. Cohort 1 compared subjects receiving PF614 and OxyContin OP with and without naltrexone blockade. Naltrexone is an opioid blocker to prevent opioids from attaching to the opioid receptors, preventing the effect of the opioid medication such as pain relief, feeling of euphoria or respiratory depression. The single ascending dose study also compared the release of oxycodone from PF614 under both fasted and fed conditions at the highest dose of PF614 evaluated, 200 mg. The pharmacokinetics of the prodrug fragments was also evaluated. In addition, this study instructed as to the “conversion efficiency” of the PF614 prodrug to oxycodone, with respect to OxyContin.
Pharmacokinetic Analyses
The shape of the plasma concentration versus time curve of oxycodone was similar following administration of OxyContin OP (oxycodone extended release) and PF614. The efficiency of conversion for PF614 to oxycodone was determined to be approximately 86%. A PF614 dose of 50 mg yields oxycodone exposure comparable to a 20.01 mg dose of OxyContin, indicating a potency ratio of 0.40. This data has allowed us to match doses of PF614 to those of commercially available OxyContin OP.
Safety
A total of 64 subjects were included in this study, of which 23 (35.9%) experienced 47 treatment-emergent adverse events, or TEAEs. The majority of TEAEs were either gastrointestinal disorders or nervous system disorders with no deaths, serious adverse events, or severe TEAEs. Additionally, there were no discontinuations due to study drug-related adverse events. Over half of TEAEs were study drug related, but they were mostly mild in severity. The three TEAEs that were moderate in severity were nephrolithiasis, or kidney stones, nausea, and vomiting, with the nausea and vomiting being study drug related. Comparing safety data across cohorts, the data indicated that dose, naltrexone, and fed/fasted state had no clinically relevant effect on the safety profile of PF614. PF614 was generally well tolerated at doses up to 200 mg in healthy subjects.
| 14 |
PF614-102 Phase 1b Clinical Trial
A Phase 1b study was conducted by ICON (formerly PRA Health Sciences) with Dr. M Johnston as Principal investigator. This was a 2-part study comprised of a MAD study (Part A) and a comparative bioavailability/bioequivalence and food effect study (Part B) in healthy subjects. Part A treated a total of 24 subjects and utilized a randomized, open-label, MAD design with up to 3 separate dose groups of 8 subjects per group. Within each dose group, subjects were randomized to receive either PF614 (n=6) or OxyContin (n=2). Subjects received repeated BID doses, planned to be administered every 12 hours (q12h) over a 5-day period, for a total of 9 doses. PF614 doses were 50 mg, 100 mg, and 200 mg, which were expected to be approximately equivalent to the 20 mg, 40 mg, and 80 mg OxyContin doses, for Dose Groups 1, 2, and 3, respectively. Serial PK sampling was performed for the first day/first dose (Day 1) and for the last day/last dose (Day 5). Only trough PK samples were taken within 30 minutes prior to the morning dose on Days 2, 3, and 4. Safety assessments, including regular assessments of adverse events (AEs), vital signs (pulse rate, blood pressure, respiratory rate, and oxygen saturation [SpO2]), clinical laboratory tests, 12-lead electrocardiograms (ECGs), and cardiac telemetry were monitored throughout the study. Subjects were monitored for hypotension, hypopnea, apnea, and oxygen desaturation.
Part B treated a total of 60 subjects and utilized an open-label, single-dose, randomized, 4-way crossover design. Subjects were randomized to receive each of the following single oral doses of study drugs in a Williams design crossover manner (1 at each treatment period):
● Treatment A: 100 mg PF614, administered under fasted conditions (hereafter referred to as 100 mg PF614, fasted)
● Treatment B: 100 mg PF614, administered under fed conditions (high-fat breakfast) (hereafter referred to as 100 mg PF614, fed)
● Treatment C: 40 mg OxyContin, administered under fasted conditions (hereafter referred to as 40 mg OxyContin, fasted)
● Treatment D: 40 mg OxyContin, administered under fed conditions (high-fat breakfast) (hereafter referred to as 40 mg OxyContin, fed)
Each treatment was separated by a washout interval of 5 days. Serial PK sampling was performed after each study drug administration up to 120 hours postdose. Safety including regular assessments of AEs, vital signs (pulse rate, blood pressure, respiratory rate, and SpO2 ), clinical laboratory tests, and 12-lead ECGs were monitored. Subjects were monitored for hypotension, hypopnea, apnea, and oxygen desaturation.
Pharmacokinetics
Part A: The shape of the plasma concentration versus time curve of oxycodone was similar following administration of PF614 and OxyContin (oxycodone extended release). Oxycodone plasma exposure (Tmax, Cmax,ss and AUCtau) were assessed and PF614 showed similar trends as OxyContin following administration of multiple oral BID doses. Due to the small sample number for OxyContin some PK parameters could not be calculated. Trough concentrations of oxycodone were generally similar from Day 2 through Day 4, suggesting that subjects achieved steady state after repeated oral BID dosing of PF614 and OxyContin at all dose levels.
Part B: A total of 57 subjects were included in the PK analyses. The data for Cmax, AUC0-t, and AUC0-inf of oxycodone post 100 mg PF614 versus 40 mg OxyContin dosing under fasted and fed conditions were completely contained within the standard bioequivalence limits of 80% to 125%. Therefore, it was concluded that 100 mg PF614 was bioequivalent to 40 mg OxyContin under both fasted and fed conditions.
Safety
PF614 was generally safe and well-tolerated following oral administration of 50 mg, 100 mg, or 200 mg PF614 BID for 5 days. There was no apparent difference in the safety profile of single oral doses of 100 mg PF614 when administered in the fasted or fed state or between PF614 and OxyContin when administered in the fasted and fed state. PF614 was generally safe and well-tolerated following single and multiple oral doses under naltrexone blockade.
| 15 |
PF614-103 Intranasal Human Abuse Potential Clinical Trial
PF614-103 was a randomized, double-blind, placebo- and active-controlled, 3-way crossover study to evaluate the abuse potential and pharmacokinetics of intranasally administered PF614, relative to crushed oxycodone IR tablets and placebo, in non-dependent recreational opioid users conducted by Lotus Clinical Trials LLC through Ohio Clinical Trials, Inc with Principal investigator, Dr. G. Apseloff.
The study consisted of 4 phases: Screening, Qualification, Treatment, and Follow-up. Subjects were randomized to receive PF614 100mg or crushed oxycodone 40 mg intranasally. The primary objective of the study was to evaluate the abuse potential of PF614 relative to crushed oxycodone immediate-release (IR) tablets and placebo following intranasal administration in non-dependent recreational opioid users (n=26), with the primary pharmacodynamic endpoint being the maximum effect (Emax) for Drug Liking (“at this moment”) measured up to 24 hours after dosing using a visual analogue scale (VAS). The secondary objectives of the study were to evaluate the pharmacokinetic profile of PF614 relative to crushed oxycodone IR tablets following intranasal administration, to evaluate the safety of PF614 following intranasal administration.
In the study, PF614 powder produced a statistically significant lower peak “drug liking” (Emax) when compared with intranasal crushed IR oxycodone (p = 0.0133) using the full modified completer population in a 3-period crossover of PF614 vs. crushed oxycodone and placebo. Furthermore, in a first period analysis of initial impressions of each drug, a statistically significant difference was noted between PF614 (n=8) and crushed IR oxycodone (n=10) (p = 0.0175), even with this smaller cohort of subjects.
Statistically significant differences in peak effects (Emax) between PF614 and crushed IR oxycodone intranasal were also demonstrated for the secondary endpoint of “take drug again,” also using a first period analysis (p < 0.0001).
The intranasal HAP study was designed to test if known recreational drug users “liked” the product and is critical for labeling claims for new drugs in this class. The primary measure in this study, “drug liking,” is recommended by the FDA in their Guidance on “Assessment of Abuse Potential of Drugs.” This measure is known to correlate with a drug’s potential for abuse. The results demonstrated that inhaled powdered PF614 had significantly lower drug liking than inhaled crushed IR oxycodone.
PF614-104 Oral Human Abuse Potential Clinical Trial
PF614-104 was a randomized, double-blind, placebo- and active-controlled, 5-way crossover study to evaluate the abuse potential and pharmacokinetics of orally administered PF614, relative to oxycodone IR tablets and placebo, in non-dependent recreational opioid users conducted by DVCR, Kansas.
The study consisted of 4 phases: Screening, Qualification, Treatment, and Follow-up. Subjects were randomized to receive PF614 50, 100 and 200 mg, oxycodone 40 mg or placebo orally. The primary objective of the study was to evaluate the abuse potential of PF614 relative to oxycodone immediate-release (IR) tablets and placebo following oral administration in non-dependent recreational opioid users (n=28), with the primary pharmacodynamic endpoint being the maximum effect (Emax) for Drug Liking (“at this moment”) and “Take Drug Again” Emax (Secondary endpoint) measured up to 24 hours after dosing using a visual analogue scale (VAS). The secondary objectives of the study were to evaluate the pharmacokinetic profile of PF614 relative to oxycodone IR tablets and to evaluate the safety of PF614.
PF614 produced statistically lower effects than oxycodone, the lowest dose p<0.0001, and statistically significant overall “Drug Liking” at both the low and the mid doses p<0.0001 and p=0.0025, respectively. PF614 took a significantly lower median time to reach Emax for “Drug Liking” than oxycodone at all three dose levels, which is highly important for reducing drug abuse. Similar findings were noted with a second endpoint “Take Drug Again”. The secondary endpoint was met at both the low and mid dose of PF614 with highly significant values of p<0.001 and p=0.0038, respectively, and was numerically lower than comparator even at double the dose, demonstrating that recreational users would be less motivated to abuse PF614 compared to immediate release oxycodone.
In November 2022, we received written guidance from the FDA that an acute pain indication may be appropriate for PF614. The FDA guidance, while not binding, states that our proposed clinical development approach of conducting at least two adequate and well-controlled clinical trials in two different pain models comparing PF614 to a placebo and to another immediate release (IR) opioid, such as IR oxycodone, appears reasonable to support a new drug application for PF614 for an acute pain indication. The FDA guidance also provides additional guidance with respect to the non-clinical studies and clinical trials planned by us. The clinical development pathway of PF614 for an acute pain indication may reduce the development timeline and be more cost-effective than initially pursuing a chronic pain indication for PF614.
| 16 |
PF614-201 Time of Onset clinical study
PF614-201 was a randomized, double-blind, placebo-controlled study of PF614 50 and 100 mg to evaluate the onset of analgesia following administration of a single oral dose of PF614 in healthy male subjects in an experimental pain model (cold pressor test [CPT]). This study was initiated in September of 2023 and enrolled 16 subjects. The Treatment Phase consisted of 2 treatment periods; PF614 50 mg or 100 mg versus placebo. The CPT, pharmacodynamic (PD), and safety assessments were conducted prior to dosing and for six hours after each study drug administration. This study was the first to successfully demonstrate the efficacy of PF614. Data was reported December 2023, stating the time-of-onset of pain relief from both doses of PF614 was identified, and PF614 did decrease the intensity of pain.
Next Steps
An End of Phase 2 regulatory meeting request for PF614 IND 116794 was submitted on October 13, 2023, with a meeting was held on January 30, 2024. The meeting clarified the non-clinical and clinical Phase 3 study plans for the further development of PF614 which are expected to initiate in mid-2024.
PF614-MPAR
We initiated a Phase 1 study that is evaluating PF614-MPAR in study entitled “A Single Dose, 2 Part Study to Evaluate the Pharmacokinetics of Oxycodone and PF614, when PF614 Solution is Co-Administered with nafamostat, as an Immediate Release Solution and/or Extended Release (ER) Capsule Formulations in Healthy Subjects:” We are clinically testing MPAR® in partnership with Quotient Sciences, using its integrated Translational Pharmaceutics® platform to search for a PF614-MPAR formulation that allows conversion into oxycodone within the prescribed dose range but reduces conversion to oxycodone at higher than prescribed dose levels in an overdose scenario.
PF614-MPAR-101 Phase 1 Clinical Trial
The primary objectives of the Phase 1 study are to assess the pharmacokinetics of oxycodone, when PF614 solution is administered alone and with nafamostat as an immediate release solution and/or extended-release capsule prototypes. The study is designed to aid in the selection of the optimal nafamostat formulation and dose to combine with PF614 in order to provide oxycodone when a prescribed dose is taken yet attenuate the maximum plasma concentration (Cmax) and the area under the concentration time curve (AUC) of oxycodone when more than the prescribed PF614-MPAR dose is taken. Initial data was reported in May 2022 that demonstrated nafamostat administer in combination with PF614 in a simulated overdose situation reduced the release of oxycodone from PF614 as designed. We completed the clinical portion of Part A of this study in December 2022, with the identification of an optimal drug product formulation. A second, Part B was initiated in January 2023 to test the overdose protection of the selected formulation by administering an escalating number of dose units to a group of healthy subjects. Enrollment was completed in March 2023.
Part B was a dose escalation study of PF614 25 mg alone, or increasing dose units of PF614-MPAR 25 mg (PF614 25 mg with 1 mg formulated nafamostat), and enrolled 6 to 8 healthy subjects in each cohort. PF614-MPAR 25 mg was delivered at 1, 2, 3, 5, and 8 doses simultaneously, with safety and PK endpoints. The plasma PK of oxycodone released from PF614 or PF614-MPAR was measured and compared to prior data where PF614 was delivered up to 200 mg alone. Additionally, the PK of parent PF614 and metabolic fragments were measured.
The results of the study successfully demonstrated that up to 2 dose units of PF614-MPAR delivered oxycodone at the same level as was derived from PF614 without MPAR®. At 3 units of PF614-MPAR the amount of oxycodone delivered was reduced compared to a 75 mg dose of PF614. At 8 dose units there was a significant decrease (p<0.00333) in the maximal oxycodone plasma concentration (Cmax) as compared to that delivered from unprotected PF614 200 mg. In addition, delivering 2 doses of PF614-MPAR sequentially, 12 hours apart did not affect the release of oxycodone from the 2nd dose. Data from this study was reported in May 2023.
| 17 |
PF614-MPAR was granted Breakthrough Therapy designation by the FDA in January 2024. A Type D meeting request to discuss the non-clinical program for PF614-MPAR was submitted in 2023 and responses to questions were received in February 2024.
Competition
The life sciences industry is characterized by rapidly advancing technologies, intense competition, and a strong emphasis on proprietary products. We expect to face competition from a number of sources, including pharmaceutical and biotechnology companies, generic drug companies, drug delivery companies, and academic and research institutions. Most of these existing and potential competitors have significantly greater financial and other resources than we do.
The key competitive factors that are expected to affect the development and commercial success of our product candidates include safety and tolerability, the ability of our product candidates to limit human abuse potential, bioavailability and therapeutic efficacy of our product candidates, market indications and convenience of dosing and distribution. PF614 will also face competition from commercially available generic and branded extended-release and long-acting opioid drugs other than oxycodone, including, but not limited to, fentanyl, hydromorphone, oxymorphone, and methadone, as well as opioids that are currently in clinical development.
We believe that obtaining an abuse-deterrent label through the FDA for our prodrugs would provide us with a significant competitive advantage. There are only four commercially available (in the United States) opioid drugs for chronic pain relief that have an abuse-deterrent label. These drugs are MorphaBond™ ER, marketed by Daiichi Sankyo, OxyContin® ER and Hysingla® ER, both of which are marketed by Purdue Pharma, LP, and Collegium Pharmaceutical, Inc.’s XTampza®ER. However, obtaining an abuse-deterrent label involves a lengthy and complicated process with no certainty of success. We believe abuse-deterrent opioids represent a therapeutic option to maximize pain relief in patients for whom opioid analgesia is indicated, while reducing the risks of abuse and diversion.
A number of other companies including, but not limited to, Pfizer Inc., Daiichi Sankyo, Endo Health Solutions, Nektar Therapeutics, Teva Pharmaceutical, Inc., Egalet Ltd., KemPharm Inc., Elysium Therapeutics Inc., and Acura Pharmaceutical, have either extended-release or abuse-deterrent products in various stages of development. Other companies offer products indicated for chronic, severe, long-term pain with various delivery technologies, but these products do not have abuse-deterrent claims on their labels. Vertex has recently announced a non-opioid pain product that inhibits NaV1.8 and has entered Phase 3 development.
We do not believe there are other companies developing products that have an overdose mechanism similar to our MPAR® technology.
Intellectual Property
Our commercial success depends in part on our ability to obtain and maintain proprietary protection for product candidates and any of our future product candidates, novel discoveries, product development technologies, and know-how; to operate without infringing on the proprietary rights of others; and to prevent others from infringing our proprietary rights. Our policy is to seek to protect our proprietary position by, among other methods, filing or in-licensing United States and foreign patents and patent applications related to our proprietary technology, inventions, and improvements that are important to the development and implementation of our business. We also rely on trademarks, trade secrets, know-how, continuing technological innovation, and potential in-licensing opportunities to develop and maintain our proprietary position.
In August 2020, EBIR entered into a Technology Transfer Agreement with Mucokinetica to acquire its intellectual property and all assets associated with the inhaled nafamostat program. Specifically, EBIR acquired Patent EP2124926B1 and all data and assets associated with the development and expansion of the inhaled nafamostat program. These assets included COVID-19 and cystic fibrosis drug targets in development.
| 18 |
In consideration for this intellectual property, Mucokinetica received a 1% equity ownership in EBIR, and its founders, Roderick Hall and Peter Cole, entered into Consulting Agreements with EBIR. The Consulting Agreements were subsequently terminated by Messrs. Hall and Cole.
Patents and Patent Applications
We own numerous patents and applications in the United States and significant commercial markets, such as Europe, China, and Japan, relating to our product candidates currently in development, as well as other product candidates that may be developed in the future. These patents and applications are projected to expire between 2028 and 2042, subject to any patent term adjustment or extension that might be available in a particular jurisdiction. A table of the key patent families and their natural or projected expiry dates is presented below.
| Countries of Filings* | Natural or Projected Expiry Date | |||
| TAAP and MPAR Patents and Applications for Opioids | ||||
| Compositions Comprising Enzyme-Cleavable Ketone-Modified Opioid Prodrugs and Optional Inhibitors Thereof | U.S., Australia, Brazil, Canada, China, Europe, Hong Kong, Israel, India, Japan, Mexico, Russia | 2030 | ||
| Compositions Comprising Enzyme-Cleavable Opioid Prodrugs and Inhibitors Thereof | U.S. | 2030 | ||
| Compositions Comprising Enzyme-Cleavable Oxycodone Prodrugs | U.S., Australia, Brazil, Canada, China, Europe, Hong Kong, Israel, India, Japan, Russia | 2032 | ||
| Compositions Comprising Enzyme-Cleavable Prodrugs and Controlled Release Nafamostat and Methods of Use Thereof | U.S., PCT, Taiwan | 2042 | ||
| Active Agent Prodrugs with Heterocyclic Linkers | U.S., Australia, Brazil, Canada, China, Europe, Hong Kong, Israel, India, Japan, Russia | 2032 | ||
| Enzyme-Cleavable Methadone Prodrugs and Methods of Use Thereof | U.S., PCT, Europe, Brazil, China, Japan, Korea, Canada, Mexico, Australia, India, Israel | 2042 | ||
| Nafamostat Patents and Applications | ||||
| Methods of Treating Coronavirus Infections and COVID-19 | U.S., Canada, Europe | 2041 | ||
| Oral formulations of Nafamostat | U.S., PCT, Taiwan, Europe, Brazil, China, Japan, Korea, Canada, Mexico, Australia, India, Israel | 2042 | ||
| Methods of Treating Respiratory Diseases with Mucostasis | Germany, France, Italy, United Kingdom | 2028 | ||
| TAAP and MPAR Patents and Applications for Amphetamines | ||||
| Compositions Comprising Enzyme-Cleavable Amphetamine Prodrugs and Inhibitors Thereof | U.S., Europe | 2031 | ||
| Compositions Comprising Enzyme-Cleavable Amphetamine Prodrugs and Inhibitors Thereof | U.S., Europe, Hong Kong | 2040 |
*”Europe” refers to patent applications filed in, and patents issued by, the European Patent Office (“EPO”), which can provide the basis for rights in multiple countries that are members of the European Patent Convention.
| 19 |
While we seek broad coverage under our existing patent applications, there is always a risk that an alteration to the products or processes may provide sufficient basis for a competitor to avoid infringing our patent claims. In addition, patents, if granted, expire and we cannot provide any assurance that any patents will be issued from our pending or any future applications or that any potentially issued patents will adequately protect our product candidates.
Individual patents extend for varying periods depending on the date of filing of the patent application or the date of patent issuance and the legal term of patents in the countries in which they are obtained. Generally, patents issued for regularly filed applications in the United States are granted a term of 20 years from the earliest effective non-provisional filing date. In addition, in certain instances, a patent term can be extended to recapture a period due to delay by the United States Patent and Trademark Office (“USPTO”) in issuing the patent as well as a portion of the term effectively lost as a result of the FDA regulatory review period. However, as to the FDA component, the restoration period cannot be longer than five years and the total patent term including the restoration period must not exceed fourteen years following FDA approval. The duration of foreign patents varies in accordance with provisions of applicable local law, but typically is also 20 years from the earliest effective non-provisional filing date. However, the actual protection afforded by a patent varies on a product-by-product basis, from country to country, and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country, and the validity and enforceability of the patent.
Our commercial success will also depend in part on not infringing upon the proprietary rights of third parties. It is uncertain whether the issuance of any third-party patent would require us to alter our development or commercial strategies for our products or processes, or to obtain licenses or cease certain activities. Our breach of any license agreements or failure to obtain a license to proprietary rights that we may require to develop or commercialize our future products may have an adverse impact on us. If third parties prepare and file patent applications in the United States that also claim technology to which we have rights, we may have to participate in interference or derivation proceedings in the USPTO to determine priority of invention. For more information, please see “Risk Factors—Risks Related to Our Intellectual Property.”
TAAP and MPAR® Patents and Applications for Opioids
Following our merger with Signature, we became the owner of patent families that include several granted U.S. patents, as well as granted patents and pending patent applications in numerous foreign jurisdictions, including Australia, Brazil, Canada, China, Europe, India, Japan, and Russia, relating to chemically modified opioids, such as oxycodone, methadone, and hydromorphone, covalently linked using specific linkers to a gastrointestinal enzyme-cleavable moiety and pharmaceutical compositions containing these modified opioids, pharmaceutical compositions containing these modified opioids and a gastrointestinal enzyme inhibitor, and methods of using the same to treat pain. Three of these patent families are directed to ketone containing opioids and cover PF614 and PF614-MPAR and certain methadone TAAP product candidates that are still in the discovery phase. These three families contain issued patents in the United States and certain foreign jurisdictions, including Australia, Brazil, Canada, China, Europe, India, Japan, and Russia and expire between 2030 and 2032, subject to any applicable patent term extension that might be available in a jurisdiction. We also own pending United States, Patent Cooperation Treaty (PCT), and Taiwan applications directed to oral formulations of PF614-MPAR, which if pursued and issued would expire in 2042, subject to any potential patent term adjustment or extension that may be available in a jurisdiction. We also own one patent family that includes granted patents in the United States, as well as granted patents and pending patent applications in numerous foreign jurisdictions, including Australia, Brazil, Canada, China, Europe, India, Japan, and Russia, relating to chemically modified ketone-containing agents, such as oxycodone, methadone, and hydromorphone, covalently linked using specific linkers to a gastrointestinal enzyme-cleavable moiety, pharmaceutical compositions containing these modified ketone-containing agents, pharmaceutical compositions containing these modified ketone-containing agents and a gastrointestinal enzyme inhibitor, and methods of using the same to treat pain, would cover certain methadone TAAP product candidates that are still in discovery phase and expire in 2032. While we own these patent families, we have not updated records in the various patent offices to reflect our ownership of these patent families. Failure to update such ownership may result in an innocent purchaser potentially acquiring rights in such patents that are adverse to our interests. Furthermore, as noted above, we have not obtained assignments for certain patent applications relating to abuse-resistant amphetamines.
| 20 |
We believe that one patent covering PF614 will be eligible for up to five years of patent term extension in the United States and intend to pursue such extension. In addition to patent exclusivity until at least 2032, under the provisions of the Hatch-Waxman Act, upon any approval in the United States, we believe that PF614 will be eligible for five-year New Chemical Entity, or NCE, regulatory exclusivity, during which time no 505(b)(2) New Drug Application, or NDA, or Abbreviated New Drug Application, or ANDA, can be approved that contains the same active moiety as the chemical entity in the PF614 NDA. In addition, if an ANDA or 505(b)(2) applicant were to file its application referencing the NDA for PF614 before expiration of our formulation patent and the applicant asserted that the patent is invalid or would not be infringed, it may be subject to additional waiting periods prior to the FDA’s approval (including a statutory thirty-month stay, starting at the end of the five-year NCE regulatory exclusivity period, if we sue for infringement, or a shorter period if the patent expires of there are certain settlements or judicial decisions in the patent litigation) and may ultimately be required to wait until the natural expiration of our compositions patents if the patents are found to be valid and infringed by the challenging applicant. For more information please see “—Patents and Patent Applications.”
Nafamostat Patents Applications
We own pending applications in the U.S., Canada and Europe directed to the use of orally administered nafamostat for the treatment of infections caused by coronaviruses, including COVID-19, and pending United States, PCT, and Taiwan patent applications directed to oral formulations of nafamostat. We intend to pursue these applications in the United States and other significant commercial markets and any patents that may be issued would expire in 2041 and 2042, respectively, subject to any applicable patent term adjustment or extension in a particular jurisdiction. Additionally, we acquired one European patent from Mucokinetica Ltd. that is directed to the use of certain compounds, including nafamostat, for the manufacture of a medicament for the treatment of respiratory diseases with mucostasis or poor mucus clearance. This patent was validated in Germany, France, Italy, and the United Kingdom and expires in 2028, subject to any applicable patent term extension that might be available in Europe Union or United Kingdom. Currently, we do not have any issued patent or pending application directed to methods of treating infections caused by coronaviruses, including COVID-19, with inhaled nafamostat, but intend to file pending applications upon development of a suitable inhalation formulation of nafamostat. We believe that one patent covering nafamostat will be eligible for up to five years of patent term extension in the United States and Europe and intend to pursue such extension. In addition to patent exclusivity, under the provisions of the Hatch-Waxman Act, upon any approval in the United States, we believe that nafamostat will be eligible for five-year NCE regulatory exclusivity, during which time no 505(b)(2) NDA or ANDA can be approved that contains the same active moiety as the chemical entity in the nafamostat NDA. In addition, if an ANDA or 505(b)(2) applicant were to file its application referencing the NDA for nafamostat before expiration of our use patent and the applicant asserted that the patent is invalid or would not be infringed, it may be subject to additional waiting periods prior to the FDA’s approval (including a statutory thirty-month stay, starting at the end of the five-year NCE regulatory exclusivity period, if we sue for infringement, or a shorter period if the patent expires of there are certain settlements or judicial decisions in the patent litigation) and may ultimately be required to wait until the natural expiration of our compositions patents if the patents are found to be valid and infringed by the challenging applicant. For more information please see “—Patent and Patent Applications.”
TAAP and MPAR® Patents and Applications for Amphetamines
Following the merger with Signature, we became the owner of one patent family that includes pending applications in the United States and numerous European foreign jurisdictions relating to chemically modified amphetamines covalently linked to a gastrointestinal enzyme-cleavable moiety, pharmaceutical compositions containing the modified amphetamines, pharmaceutical compositions containing the modified amphetamines and a gastrointestinal enzyme inhibitor and methods of using the same to treat a subject. While we own this patent family, we have not updated the records in the various patent offices to reflect our ownership of this patent family. Failure to update such ownership may result in an innocent purchaser potentially acquiring rights in such patents that are adverse to our interests. In addition, we own pending United States and European patent applications directed to pharmaceutical compositions containing chemically modified amphetamines covalently linked to a gastrointestinal enzyme-cleavable moiety and a trypsin inhibitor and methods of using the same to treat a subject. We have not obtained assignments from all of the inventors of these applications to date, which could negatively impact our ability to pursue or enforce this application. If issued, these patent applications would expire between 2031 and 2040, subject to any applicable patent term adjustment or extension that might be available in a jurisdiction.
| 21 |
Trademarks and Trade Secrets
We intend to pursue trademark registrations in the United States and other significant commercial markets for our product candidates as they progress through clinical development. We received registration of our trademark for MPAR on May 16, 2023.
Furthermore, we rely upon trade secrets, know-how, continuing technological innovation, and potential in-licensing opportunities to develop and maintain our competitive position. We seek to protect our proprietary information, in part, using confidentiality and invention assignment agreements with our commercial partners, collaborators, employees, and consultants. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with an employee or a third party. These agreements may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our commercial partners, collaborators, employees, and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Manufacturing and Supply
We do not currently own or operate manufacturing facilities for the production of clinical or commercial quantities of our product candidates. Our drug substance and drug products are manufactured for us by contract manufacturing organizations, or CMOs, to our specifications. Any manufacturing problem or the loss of a CMO could be disruptive to our operations.
Our lead product candidate, PF614, is a small molecule opioid prodrug. As such, it is a controlled substance, regulated by the Drug Enforcement Administration (“DEA”) and state-controlled substance authorities. Our CMOs will be required to be registered with DEA and will be responsible for obtaining adequate quota to manufacture and otherwise handle controlled substances.
We currently engage third parties to provide clinical supplies of PF614 and nafamostat. We also currently engage a CMO to provide drug product manufacture of PF614, PF614-MPAR and nafamostat. We currently have sufficient supplies of PF614 and nafamostat on hand for our current clinical trial needs. Any reliance on suppliers may involve several risks, including a potential inability to obtain critical materials and reduced control over production costs, delivery schedules, reliability, and quality.
Purisys
Purisys LLC manufactures PF614 and other clinical trial materials under cGMP conditions and provides stability studies with respect to our PF614 clinical trials. We do not currently have a binding written agreement with Purisys. In the event that Purisys is unable to perform the services promised under future agreements, we may be subject to unforeseen costs and delays with respect to our clinical trials and may be unable to replace the Purisys arrangements on terms as favorable to us. See “Risk Factors—We expect to be completely dependent on third parties to manufacture our product candidates, and our commercialization of our product candidates could be halted, delayed or made less profitable if those third parties fail to maintain a compliance status acceptable to the FDA or comparable foreign regulatory authorities, fail to provide to us with sufficient quantities of our product candidates or fail to do so at acceptable quality levels or prices” for more information.
Societal CDMO
Societal CDMO manufactures PF614 and other clinical trial drug products under cGMP conditions and provides stability studies with respect to our PF614 clinical trials. Societal has completed the manufacture of PF614 50 and 100 mg capsules that have been used in clinical studies PF614-102, PF614-103, PF614-104 and PF614-201. We expect to enter into additional related agreements with Societal CDMO as we manufacture future batches of PF614. In the event that Societal is unable to perform the services anticipated under future agreements, we may be subject to unforeseen costs and delays with respect to our clinical trials. See “Risk Factors—We expect to be completely dependent on third parties to manufacture our product candidates, and our commercialization of our product candidates could be halted, delayed or made less profitable if those third parties fail to maintain a compliance status acceptable to the FDA or comparable foreign regulatory authorities, fail to provide to us with sufficient quantities of our product candidates or fail to do so at acceptable quality levels or prices” for more information.
| 22 |
Government Grants
We have received funding under federal grant award programs through governmental agencies, such as the NIH and NIDA. For the year ended December 31, 2023, we received federal grant funding totaling $2.2 million, $1.3 million from NIH related to the Phase 1 clinical trial for PF614-MPAR and $0.9 million from NIDA for preclinical development of our opioid use disorder-MPAR® technology. Current remaining funding under approved grants totaled $2.2 million as of December 31, 2023, covering the period through August 2024. We may apply for additional grant funding from these or similar governmental agencies in the future.
GEM Facility
Pursuant to the GEM Agreement, we are entitled to draw down up to $60 million of gross proceeds (“Aggregate Limit”) from GEM Global in exchange for shares of our common stock, subject to meeting the terms and conditions of the GEM Agreement. This equity line facility is available for a period of 36 months from the closing date of the Merger. A draw down is subject to limitations on the amount that is drawn under the facility and must comply with certain conditions precedent including the listing of our shares on a principal market (which includes Nasdaq), having the necessary number of shares that are issuable pursuant to the draw down registered under an effective registration statement, and other notice and timing requirements. Upon our valid exercise of a draw down, pursuant to delivery of a notice and in accordance with other conditions, GEM Global is required to pay, in cash, a per-share amount equal to 90% of the average closing bid price of the shares of our common stock recorded by Nasdaq during the 30 consecutive trading days commencing on the first trading day that is designated on the draw down notice. In no event may our draw down requests exceed 400% (“Draw Down Limit”) of the average daily trading volume for the 30 trading days immediately preceding the date we deliver the draw down notice.
Further, upon the closing of the Merger, GEM Global became entitled to a commitment fee in the form of cash or freely tradeable shares of our common stock in an amount equal to 2% of the Aggregate Limit or $1.2 million to be paid in two tranches. The commitment fees have been paid in full.
Additionally, we issued a warrant with a 36-month term at the closing of the Merger granting GYBL the right to purchase 4,608 shares of our common stock at a strike price per share equal to $1.5675, after several downward adjustments to the strike price. Any failure by us to timely transfer the shares under the warrant pursuant to GYBL’s exercise will entitle GYBL to compensation in addition to other remedies. The number of shares underlying the warrant as well as the strike price is subject to adjustments for recapitalizations, reorganizations, change of control, stock split, stock dividend and reverse stock splits. The strike price is subject to adjustment for issuances of additional common shares at a price per share less than the strike price.
The GEM Agreement contains certain negative covenants restricting us from securing an equity line similar to the financing provided under the GEM Agreement and requiring prompt notice of events constituting an alternate transaction. An “alternate transaction” includes an issuance of common stock at a price less than the then current market price, an “at-the-market” offering of securities, and an issuance of options, warrants, or similar rights of subscription or the issuance of convertible equity or debt securities. See “Risks Related to Our Business, Financial Condition and Capital Requirements” for additional information.
Finally, pursuant to the terms of the GEM Agreement, we are required to indemnify GEM Global for any losses it incurs as a result of a breach by us or of our representations and warranties and covenants under the GEM Agreement or for any misstatement or omission of a material fact in a registration statement registering those shares pursuant to the GEM Agreement. Also, GEM Global is entitled to be reimbursed for legal or other costs or expenses reasonably incurred in investigating, preparing, or defending against any such loss. To date, we have not raised any capital pursuant to the GEM facility and we may not raise any capital pursuant to it prior to its expiration. Restrictions pursuant to the terms of our recent financings may also affect our ability to use the GEM Facility.
| 23 |
Government Regulation
In the United States, pharmaceutical products are subject to extensive regulation by the FDA, and those pharmaceutical products that are controlled substance are also subject to extensive regulation by the DEA. The Federal Food, Drug, and Cosmetic Act (the “FDC Act”), the Controlled Substances Act (“CSA”), and other federal, state, and local statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, prescribing, dispensing, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Pharmaceutical products used for the prevention, treatment, or cure of a disease or condition of a human being are subject to regulation under the FDC Act. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as clinical hold, FDA refusal to approve pending NDAs, revocation of licensing authority, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, and criminal prosecution.
The FDA Drug Approval Process
FDA approval is required before any new drug can be marketed. A new drug is one not generally recognized by experts qualified by scientific training and experience, as safe and effective for its intended use. The process of drug development is complex and lengthy. The activities undertaken before a new pharmaceutical product may be marketed in the United States generally include, but are not limited to, preclinical studies; submission to the FDA of an IND, which must become active before human clinical trials may commence; adequate and well-controlled human clinical trials to establish the safety and efficacy of the product; submission to the FDA of an NDA; filing of the NDA by FDA; satisfactory completion of an FDA pre-approval inspection of the clinical trial sites and manufacturing facility or facilities at which both the active ingredients and finished drug product are produced to assess compliance with, among other things, patient informed consent requirements, the clinical trial protocols, current Good Clinical Practices, or GCP, and GMPs; and FDA review and approval of the NDA prior to any commercial sale and distribution of the product in the United States.
Preclinical studies include laboratory evaluation of product chemistry and formulation, and in some cases, animal studies and other studies to preliminarily assess the potential safety and efficacy of the product candidate. The results of preclinical studies together with manufacturing information, analytical data, and detailed information including protocols for proposed human clinical trials are then submitted to the FDA as a part of an IND. An IND must become effective, and approval must be obtained from an Institutional Review Board (“IRB”) prior to the commencement of human clinical trials. The IND becomes effective 30 days following its receipt by the FDA unless the FDA objects to, or otherwise raises concerns or questions and imposes a clinical hold. We, the FDA, or the IRB may suspend or terminate a clinical trial at any time after it has commenced due to safety or efficacy concerns or for commercial reasons. In the event the FDA imposes a clinical hold, the IND sponsor must address any outstanding FDA concerns or questions to the satisfaction of the FDA before clinical trials can proceed or resume.
Human clinical trials are typically conducted in three sequential phases that may sometimes overlap or be combined:
In Phase 1, the initial introduction of the drug into patients, the product is tested to assess safety, dosage tolerance, metabolism, pharmacokinetics, pharmacological actions, side effects associated with drug exposure, and to obtain early evidence of a treatment effect if possible. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, determine optimal dose and regimen, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain additional information about clinical effects and confirm efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the product. In most cases, the FDA requires two adequate and well-controlled Phase 3 clinical trials to demonstrate the safety and efficacy of the drug. In rare instances, a single Phase 3 trial may be sufficient when either (1) the trial is a large, multicenter trial demonstrating internal consistency and a statistically very persuasive finding of a clinically meaningful effect on mortality, irreversible morbidity, or prevention of a disease with a potentially serious outcome and confirmation of the result in a second trial would be practically or ethically impossible or (2) the single trial is supported by other confirmatory evidence.
| 24 |
In addition, the manufacturer of an investigational drug in a Phase 2 or Phase 3 clinical trial for a serious or life-threatening disease is required to make available, such as by posting on its website, its policy on evaluating and responding to requests for expanded access to such investigational drug.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing and distribution of the product may begin in the United States. The NDA must include the results of all preclinical, clinical, and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture, and controls. The cost of preparing and submitting an NDA is substantial. The submission of most NDAs is additionally subject to a substantial application user fee, currently exceeding $3.1 million for Fiscal Year 2022. Under an approved NDA, the applicant is also subject to an annual program fee, currently exceeding $330,000. These fees typically increase annually. Under limited circumstances, an applicant may be exempt from or seek a waiver of the application fee requirement.
The FDA has 60 days from its receipt of an NDA to determine whether the application will be filed based on the FDA’s determination that it is adequately organized and sufficiently complete to permit substantive review. Once the submission is filed, the FDA begins an in-depth review. The FDA has agreed to certain performance goals to complete the review of NDAs. For a standard review, the goal for review of a new molecular entity (“NME”) is ten months from the date the FDA files the NDA, while the goal for review of a non-NME is ten months from the date of receipt of the NDA. For an NDA that has received a priority review designation from the FDA, the goal for review of an NME is six months from the date the FDA files the NDA, while the goal for review of a non-NME is six months from the date of receipt of the NDA. An NDA can receive a priority review designation when the FDA determines the drug has the potential to treat a serious or life-threatening condition and, if approved, would be a significant improvement in safety or effectiveness compared to available therapies. The review process for both standard and priority reviews may be extended by the FDA for three or more additional months to consider certain late-submitted information, or information intended to clarify information already provided in the NDA submission.
The FDA may also refer applications for novel drug products, as well as drug products that present difficult questions of safety or efficacy, to be reviewed by an advisory committee—typically a panel that includes clinicians, statisticians, and other experts—for review, evaluation, and a recommendation as to whether the NDA should be approved. The FDA is not bound by the recommendation of an advisory committee, but generally follows such recommendations. Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug product is manufactured. The FDA will not approve the product unless compliance with cGMP is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the claimed indication.
After the FDA evaluates the NDA and completes any clinical and manufacturing site inspections, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the NDA submission and may require substantial additional testing, or information, in order for the FDA to reconsider the application for approval. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. The FDA has committed to reviewing such resubmissions in two or six months depending on the type of information included. An approval letter authorizes commercial marketing and distribution of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy (“REMS”) to help ensure that the benefits of the drug outweigh the potential risks to patients. A REMS can include medication guides, communication plans for healthcare professionals, and elements to assure a products safe use (“ETASU”). An ETASU REMS can include, but is not limited to, special training or certification for prescribing or dispensing the product, dispensing the product only under certain circumstances, special monitoring, and the use of patient-specific registries. The requirement for a REMS can materially affect the potential market and profitability of the product. Moreover, the FDA may require substantial post-approval testing and surveillance to monitor the product’s safety or efficacy.
Before approval, the FDA evaluates the results from in vitro manipulation and extraction, pharmacokinetics, and clinical human abuse potential studies to determine whether the accumulated evidence is sufficient to warrant claims of abuse deterrence. Post-marketing studies may also be required to determine whether the marketing of a product with abuse-deterrent properties results in meaningful reductions in abuse, misuse, and related adverse clinical outcomes, including addiction, overdose, and death in the post-approval setting.
| 25 |
Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing. Changes to some of the conditions established in an approved NDA, including changes in indications, product labeling, manufacturing processes, or facilities, require submission and FDA approval of a new NDA, or supplement to an approved NDA, before the change can be implemented. An NDA supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing NDA supplements as it does in reviewing original NDAs.
Section 505(b)(2) NDAs
An alternative to the NDA pathway described above is an NDA submitted under Section 505(b)(2) of the FDC Act, which enables the applicant to rely, in part, on the FDA’s prior findings in approving a similar product or published literature in support of its application. Section 505(b)(2) NDAs often provide an alternate path to FDA approval for modified formulations, new routes of administration, or new uses of previously approved products. Section 505(b)(2) permits the submission of an NDA where at least some of the information required for approval comes from studies not conducted by, or for, the applicant and for which the applicant has not obtained a right of reference. If the Section 505(b)(2) applicant can establish that reliance on the FDA’s prior findings of safety or effectiveness is scientifically appropriate, it may eliminate the need to conduct certain preclinical or clinical studies of the new product. The FDA may also require companies to perform additional studies or measurements to support the change from the approved product. The FDA may then approve the new product candidate for all, or some, of the indications for which the referenced product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant.
Fast Track Designation and Priority Review
FDA is required to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. Fast Track designation may be granted for products that are intended to treat a serious or life-threatening disease or condition for which there is no effective treatment and preclinical or clinical data demonstrate the potential to address unmet medical needs for the condition. Fast Track designation applies to both the product and the specific indication for which it is being studied. Any product submitted to FDA for marketing, including under a Fast Track designation, may be eligible for other types of FDA programs intended to expedite development and review, such as priority review.
Priority review may be granted for products that are intended to treat a serious or life-threatening condition and, if approved, would provide a significant improvement in safety and effectiveness compared to available therapies. FDA will attempt to direct additional resources to the evaluation of an application designated for priority review to facilitate the review.
Breakthrough Therapy Designation
FDA Breakthrough Therapy designation is designed to expedite the development and review of drugs that are intended to treat a serious condition where preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over available therapies. The primary intent of Breakthrough Therapy designation is to develop evidence needed to support approval as efficiently as possible. The designation provides all the features of Fast Track designation including accelerated approval and priority review along with intensive guidance involving FDA senior managers on an efficient drug development program.
Disclosure of Clinical Trial Information
Sponsors of clinical trials of FDA-regulated products, including drugs, are required to register and disclose certain clinical trial information on the website www.clinicaltrials.gov. Information related to the product, patient population, phase of investigation, trial sites and investigators, and other aspects of a clinical trial are then made public as part of the registration. Sponsors are also obligated to disclose the results of their clinical trials after completion. Disclosure of the results of clinical trials can be delayed in certain circumstances for up to two years after the date of completion of the trial. Competitors may use this publicly available information to gain knowledge regarding the progress of clinical development programs as well as clinical trial design.
| 26 |
The Hatch-Waxman Amendments
Under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Amendments, a portion of a product’s U.S. patent term that was lost during clinical development and regulatory review by the FDA may be restored. The Hatch-Waxman Amendments also provide a process for listing patents pertaining to approved products in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations (commonly known as the “Orange Book”) and for a competitor seeking approval of an application that references a product with listed patents to make certifications pertaining to such patents. In addition, the Hatch-Waxman Amendments provide for a statutory protection, known as non-patent exclusivity, against the FDA’s acceptance or approval of certain competitor applications.
Patent Term Extension
Patent Term Extension (“PTE”) in the United States can compensate for lost patent grant time during product development and the regulatory review process for a patent that covers a new product or its use. This PTE period is generally one-half the time between the effective date of an IND (falling after issuance of the patent) and the submission date of an NDA, plus the time between the submission date of an NDA and the approval of that application, provided the sponsor acted with diligence. PTEs that can be obtained are for up to five years beyond the expiration of the patent or fourteen years from the date of product approval, whichever is earlier. Only one patent applicable to an approved drug may be extended and the extension must be applied for prior to expiration of the patent. The USPTO, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration.
The term of individual patents depends upon the legal term of the patents in the countries in which they are obtained. In most countries in which we file, the patent term is 20 years from the earliest date of filing a nonprovisional patent application related to the patent. A U.S. patent also may be accorded patent term adjustment, or PTA, under certain circumstances to compensate for delays in obtaining the patent from the USPTO. In some instances, such a PTA may result in a U.S. patent term extending beyond 20 years from the earliest date of filing a non-provisional patent application related to the U.S. patent. In addition, in the United States, the term of a U.S. patent that covers an FDA-approved drug may also be eligible for a patent term extension, or PTE, which permits patent term restoration as compensation for the patent term lost during the FDA regulatory review process. The Hatch-Waxman Act permits a PTE of up to five years beyond the expiration of the patent. The length of the PTE is related to the length of time the drug is under regulatory review. PTE cannot extend the remaining term of a patent beyond a total of fourteen years from the date of product approval and only one patent applicable to an approved drug may be extended. Similar provisions are available in Europe and certain other jurisdictions to extend the term of a patent that covers an approved drug. In the future, if and when our products receive FDA approval, we expect to apply for PTEs on patents covering products eligible for PTE. We plan to seek PTEs for any of our issued patents in any jurisdiction where these are available; however, there is no guarantee that the applicable authorities, including the FDA in the United States, will agree with our assessment of whether such extensions should be granted, and if granted, the length of such extensions.
We also believe that (1) PF614 and nafamostat will be eligible for a five-year NCE regulatory exclusivity, and (2) PF614-MPAR will be eligible for a three-year clinical investigation, or CI, regulatory exclusivity, under the Hatch-Waxman Act, during which time no ANDA can be approved.
Under the Hatch-Waxman Act, patents covering the product such as patents claiming the approved composition of matter, approved methods of use, approved formulations, and approved dosing and administration shall be listed in the Orange Book, which identifies drug products approved by FDA under the FDC Act. Applicable regulatory exclusivities, such as the five-year NCE exclusivity and the three-year CI exclusivity, are also listed in the Orange Book. If an ANDA or 505(b)(2) applicant were to file its application before expiration of all patents listed in the Orange Book, it must certify whether it will either honor or challenge all the patents listed in the Orange Book. If an Orange Book listed patent is challenged and we sue the ANDA or 505(b)(2) applicant for infringement, a statutory 30-month stay of approval, started at the end of the NCE exclusivity period, will be put in place that will prohibit the FDA from finally approving the ANDA or 505(b)(2) application until the 30-months have expired or after a court has held in favor of the ANDA or 505(b)(2) applicant. The 30-month stay begins at the end of the five-year NCE exclusivity period. If the Orange Book listed patent(s) is ultimately held valid and infringed, the ANDA or 505(b)(2) applicant will not be finally approved until the Orange Book listed patent(s) expires. If a pediatric study is requested by the FDA in a Pediatric Written Request, or PWR, and we complete the pediatric study according to the terms of the PWR, all unexpired Orange Book listed exclusivities (patent or regulatory) will be extended by six months.
| 27 |
Similar provisions are available in Europe, Japan, and certain other jurisdictions to extend the exclusivity of a patent that covers an approved drug. In Europe, we believe PF614 and nafamostat will be eligible for 10 years of regulatory exclusivity from European Marketing Application, or EMA, approval. In Japan, we believe PF614 will be eligible for eight years of regulatory exclusivity from a Japanese new drug application, or J-NDA, approval.
Orange Book Listing
In seeking approval for a drug through an NDA, applicants are required to list with the FDA each patent with claims covering the applicant’s product or method of using the product. Upon approval of a drug, each of the patents identified in the application for the drug are then published in the FDA’s Orange Book. Drugs listed in the Orange Book can, in turn, be cited by potential generic competitors in support of approval of an ANDA. An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths and dosage form as the listed drug and has been shown to be bioequivalent to the listed drug. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, preclinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the listed drug and can often be substituted by pharmacists under prescriptions written for the original listed drug.
The ANDA applicant is required to certify to the FDA concerning any patents listed for the approved product in the FDA’s Orange Book. Specifically, the applicant must certify that: (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid or will not be infringed by the new product. The ANDA applicant may also elect to submit a Section VIII statement certifying that its proposed ANDA labeling does not contain (or carves out) any language regarding the patented method-of-use rather than certify to a listed method-of-use patent. If the applicant does not challenge the listed patents, the ANDA application will not be approved until all the listed patents claiming the referenced product have expired.
A certification that the new product will not infringe the already approved product’s listed patents, or that such patents are invalid, is called a Paragraph IV certification. If the ANDA applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the ANDA has been filed with and accepted by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days of the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, or a decision in the infringement case that is favorable to the ANDA applicant.
An applicant submitting an NDA under Section 505(b)(2) of the FDC Act, which permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by, or for, the applicant and for which the applicant has not obtained a right of reference, is required to certify to the FDA regarding any patents listed in the Orange Book for the approved product it references to the same extent that an ANDA applicant would.
Market Exclusivity
Market exclusivity provisions under the FDC Act also can delay the submission or the approval of certain applications. The FDC Act provides a five-year period of non-patent marketing exclusivity within the United States to the first applicant to gain approval of an NDA for a new chemical entity (“NCE”). A drug is entitled to NCE exclusivity if it contains a drug substance with no active moiety of which has been previously approved by the FDA. During the exclusivity period, the FDA may not accept for review an ANDA or a 505(b)(2) NDA submitted by another company for another version of such drug where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a Paragraph IV certification. For a drug that has been previously approved by the FDA, the FDC Act also provides three years of marketing exclusivity for an NDA, 505(b)(2) NDA, or supplement to an existing NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example, for new indications, dosages or strengths of an existing drug. This three-year exclusivity covers only the new conditions of use and does not prohibit the FDA from approving ANDAs for drugs for the original conditions of use, such as the originally approved indication. Five-year and three-year exclusivity will not delay the submission or approval of a full NDA; however, an applicant submitting a full NDA would be required to conduct or obtain a right of reference to all of the non-clinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
| 28 |
Post-Marketing Requirements
Following approval of a new product, a pharmaceutical company and the approved product are subject to continuing regulation by the FDA, including, among other things, monitoring and recordkeeping activities, reporting to the applicable regulatory authorities of adverse experiences with the product, providing the regulatory authorities with updated safety and efficacy information, product sampling and distribution requirements, and complying with promotion and advertising requirements, which include, among others, standards for direct-to-consumer advertising, restrictions on promoting drugs for uses or in patient populations that are not described in the drug’s approved labeling (known as “off-label use”), limitations on industry-sponsored scientific and educational activities and requirements for promotional activities involving the internet. Although physicians may prescribe legally available drugs for off-label uses, manufacturers may not market or promote such off-label uses. Modifications or enhancements to the product or its labeling or changes of the site of manufacture are often subject to the approval of the FDA and other regulators, who may or may not grant approval or may include in a lengthy review process.
Prescription drug advertising is subject to federal, state, and foreign regulations. In the United States, the FDA regulates prescription drug promotion, including direct-to-consumer advertising. Prescription drug promotional materials must be submitted to the FDA in conjunction with their first use. Any distribution of prescription drug products and pharmaceutical samples must comply with the U.S. Prescription Drug Marketing Act (“PDMA”), a part of the FDC Act. In addition, Title II of the Federal Drug Quality and Security Act of 2013, known as the Drug Supply Chain Security Act or the DSCSA, has imposed new “track and trace” requirements on the distribution of prescription drug products by manufacturers, distributors, and other entities in the drug supply chain. These requirements are being phased in over a ten-year period. Unless the products were packaged prior to November 27, 2018, the DSCSA requires product identifiers (i.e., serialization) on prescription drug products in order to establish an electronic interoperable prescription product system to identify and trace certain prescription drugs distributed in the United States. The DSCSA replaced the prior drug “pedigree” requirements under the PDMA and preempts existing state drug pedigree laws and regulations. The DSCSA also establishes requirements for the licensing of wholesale distributors and third-party logistic providers. These licensing requirements preempt states from imposing licensing requirements that are inconsistent with, less stringent than, directly related to, or otherwise encompassed by standards established by FDA pursuant to the DSCSA. Until FDA promulgates regulations to address the DSCSA’s new national licensing standard, current state licensing requirements typically remain in effect.
In the United States, once a product is approved, its manufacture is subject to comprehensive and continuing regulation by the FDA. The FDA regulations require that products be manufactured in specific facilities and in accordance with cGMP. cGMP regulations require among other things, quality control and quality assurance as well as the corresponding maintenance of records and documentation and the obligation to investigate and correct any deviations from cGMP. Drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and certain state agencies and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP and other laws. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance. These regulations also impose certain organizational, procedural, and documentation requirements with respect to manufacturing and quality assurance activities. NDA holders using contract manufacturers, laboratories, or packagers are responsible for the selection and monitoring of qualified firms, and, in certain circumstances, qualified suppliers to these firms. These firms and, where applicable, their suppliers are subject to inspections by the FDA at any time, and the discovery of violative conditions, including failure to conform to cGMP, could result in enforcement actions that interrupt the operation of any such product or may result in restrictions on a product, manufacturer, or holder of an approved NDA, including, among other things, recall or withdrawal of the product from the market.
| 29 |
The CSA and DEA Regulation
Our products are regulated as “controlled substances” as defined under the CSA and regulations promulgated by DEA. The law and regulations establish registration, security, recordkeeping, reporting, storage, distribution, importation, exportation, and other requirements administered by DEA.
Controlled substances are classified into five schedules: Schedule I, II, III, IV, or V, depending on the abuse potential. Schedule I substances by definition have no established medicinal use and may not be marketed or sold in the United States. A pharmaceutical product may be listed as Schedule II, III, IV, or V, with Schedule II substances considered to present the highest risk of abuse and Schedule V substances the lowest relative risk of abuse among such substances.
PF614 will be classified as a Schedule II controlled substance under the CSA and regulations because it contains oxycodone which is already regulated as a Schedule II controlled substance. Consequently, the manufacturing, shipping, storing, selling, prescribing, and dispensing of our products is subject to a high degree of regulation. Schedule II drugs are subject to the strictest requirements for registration, security, recordkeeping, and reporting. Facilities must maintain complete and accurate inventories and records of all controlled substances received, manufactured, stored, and distributed. These facilities must comply with strict security requirements to prevent diversion of drugs in their possession. Also, distribution and dispensing of these drugs are highly regulated. For example, all Schedule II drug prescriptions must be signed by a physician, presented to a pharmacist and generally limited to a 30-day supply, and may not be refilled, that is, a new prescription is required.
Annual registration is required for any facility that manufactures, distributes, imports, or exports any controlled substance. Also, practitioners and pharmacies are required to register every three years. The registration is specific to the location, activity, and controlled substance schedule. For example, separate registrations are needed for import and manufacturing, and each registration will specify which schedules of controlled substances the facility is authorized to handle. Our contract manufacturers must be registered with DEA.
In addition, the CSA establishes an annual quota system that limits the manufacturing of API and dosage forms in the United States of Schedule I and II controlled substances. First, the DEA establishes an annual aggregate quota for how much active opioid ingredients, such as oxycodone and tapentadol, may be produced in total in the United States based on the DEA’s estimate of the quantity needed to meet legitimate scientific and medicinal needs. The limited aggregate number of opioids that the DEA allows to be produced in the United States each year is allocated among individual companies, who must submit applications annually to the DEA for individual production quotas. Also, dosage form manufacturers must also request a procurement quota to acquire opioid API to manufacture dosage forms for distribution. We and our contract manufacturers must receive an annual quota from the DEA in order to produce or procure any Schedule I or Schedule II substance, including oxycodone base for use in manufacturing PF614. The DEA may adjust aggregate production quotas and individual production and procurement quotas from time to time during the year. DEA has substantial discretion in whether or not to make such adjustments. Our contract manufacturers must apply for and obtain the necessary quotas on an annual basis.
In November 2017, the DEA reduced the amount of almost every Schedule II opiate and opioid medication that may be manufactured in the United States in calendar year 2018 by 20%. In October 2018, the SUPPORT Act was enacted, which included amendments to the CSA to require that appropriate quota reductions be made after estimating potential for diversion. DEA announced that the estimate is based on rates of overdose deaths and abuse, the overall public health impact related to specific controlled substances and may include other factors as appropriate. For 2019, the DEA proposed decreased manufacturing quotas for the six most frequently misused opioids, including oxycodone, by an average of 10% as compared to the 2018 quotas. In October 2019, consistent with the SUPPORT Act, DEA proposed additional regulations to amend the manner in which the agency grants quotas to manufacturers. The proposed regulations will establish use-specific quotas, including commercial sales, product development, transfer, replacement, and packaging. To decrease the risk of diversion and increase accountability, inventory allowances will be reduced, and procurement quota certifications will be required. The DEA proposed further decreasing manufacturing quotas in 2020 for five of the six opioids (fentanyl, hydrocodone, hydromorphone, oxycodone, and oxymorphone), by an average of 28%. For 2021, the DEA decreased the aggregate quota for oxycodone by about 13% and for hydrocodone by about 10% from the final established 2020 quotas. Because PF614 is regulated as a Schedule II controlled substance, it is subject to the DEA’s aggregate, individual production, and procurement quota scheme.
| 30 |
Ordering and distribution of any Schedule I or II controlled substance are also subject to special ordering requirements under either the electronic Controlled Substance Ordering System (“CSOS”) or use of DEA Form 222s. Information regarding specific transactions are reported to DEA, and cumulative reports of such transactions are required monthly/quarterly.
The DEA also requires drug manufacturers to design and implement a system that identifies and reports suspicious orders of controlled substances. Such orders include those of unusual size, those that deviate substantially from a normal pattern, and those of unusual frequency. Manufacturers must refuse to complete any sale and report to DEA any orders for which it is unable to resolve any potential “red flags.” A compliant suspicious order monitoring system includes well-defined due diligence, “know your customer” process as well as systems to identify and monitor ordering and sales of controlled substances.
To enforce these requirements, the DEA conducts periodic inspections of registered establishments that handle controlled substances. Failure to maintain compliance with applicable requirements, especially security and recordkeeping and as manifested in loss or diversion or inability to account for all controlled substances, can result in administrative, civil, or criminal enforcement action that could have a material adverse effect on our business, results of operations, and financial condition. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate administrative proceedings to revoke those registrations. The DEA may also reduce or deny quota to manufacturing facilities based on non-compliance with these requirements. In certain circumstances, violations could result in criminal proceedings.
Individual states also independently regulate controlled substances.
Legislative and Regulatory Initiatives for Opioids
In response to widespread prescription opioid abuse, the United States government and a number of state legislatures have enacted legislation and regulations intended to fight the opioid epidemic. The number and scope of legislative and regulatory actions, particularly in the last three years, emphasize the severity of the opioid epidemic and its impact on our society. The FDA has stated that addressing prescription drug abuse is a priority and has reaffirmed that the development of abuse-deterrent opioids is a key part of that strategy.
Recent actions to address the opioid abuse epidemic include:
| ● | FDA guidance: In April 2015, the FDA adopted final guidance regarding studies and clinical trials that should be conducted to demonstrate that a given formulation has abuse-deterrent properties, how those studies and clinical trials will be evaluated, and what product labeling claims may be approved based on the results of those studies and clinical trials. The guidance describes four categories of abuse-deterrence studies and clinical trials: Categories 1, 2, and 3 consist of pre-marketing studies and clinical trials designed to evaluate a product candidate’s potentially abuse-deterrent properties under controlled conditions, while Category 4, post-marketing clinical trials and studies, assesses the real-world impact of abuse-deterrent formulations. The final guidance also provides examples of product label claims that may be made based on the results of the corresponding studies and clinical trials. | |
| ● | FDA Opioids Action Plan: In February 2016, the FDA released an action plan to address the opioid abuse epidemic and reassess the FDA’s approach to opioid medications. The FDA’s plan is part of a broader initiative led by the U.S. Department of Health and Human Services (“HHS”), to address opioid-related overdose, death, and dependence. | |
| ● | CDC Prescribing Guidelines: In November 2022, the CDC released a new Guideline for Prescribing Opioids for Pain to update their 2016 Guidelines. The new guidance includes recommendations for managing acute (duration of <1 month), subacute (duration of 1–3 months), and chronic (duration of >3 months) pain. The guideline addresses the following four areas: 1) determining whether or not to initiate opioids for pain, 2) selecting opioids and determining opioid dosages, 3) deciding duration of initial opioid prescription and conducting follow-up, and 4) assessing risk and addressing potential harms of opioid use. The guideline addresses the following four areas: 1) determining whether or not to initiate opioids for pain, 2) selecting opioids and determining opioid dosages, 3) deciding duration of initial opioid prescription and conducting follow-up, and 4) assessing risk and addressing potential harms of opioid use. |
| 31 |
| ● | FDA Drug Safety Communication: In April 2023, the FDA issued a communication that in the ongoing effort to address the nation’s opioid crisis, it was making several updates to the prescribing information of opioid pain medicines to provide additional guidance on their use. The changes include label updates addressing addiction, abuse and misuse as well as life-threatening respiratory depression, accidental ingestion, risks from concomitant use with other CNS depressants, neonatal withdrawal and opioid analgesic risk evaluation and mitigation strategy. | |
| ● | Enhanced Warnings and Safety Labeling: In March 2016, the FDA announced required enhanced warnings for immediate-release opioid pain medications related to risks of misuse, abuse, addiction, overdose, and death. Subsequently, there have been several class-wide labeling changes, including the addition of boxed warnings relating to serious risks of using certain opioids medications along with benzodiazepines and other central nervous system depressants, including alcohol (Decembers 2016); and additional information relating to the new class-wide REMS (Septembers 2018). | |
| ● | Enactment of the Comprehensive Addiction and Recovery Act (“CARA”): In 2016, the CARA was enacted to address the national epidemics of prescription opioid abuse and heroin use. Consistent with the initiatives of HHS, this legislation sought to, among other things, expand the availability of naloxone for law enforcement and other first responders; form an interagency task force to develop best practices for pain management with opioid medications; and provide resources to improve state monitoring of controlled substances, including opioids. In 2018, CARA 2.0 was introduced as follow-up legislation to limit initial prescriptions for opioids to 3 days, while exempting initial prescriptions for chronic care, cancer care, hospice or end of life care, and palliative care. | |
| ● | Enactment of the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act (“SUPPORT Act”): In November 2018, the SUPPORT Act was enacted as a comprehensive legislative response to the continuing opioid epidemic. It includes a number of measures directed towards regulation and improvement of treatment for substance use-disorder and increased coverage by CMS of medically assisted treatment options. In addition, the SUPPORT Act requires HHS to report to Congress on existing barriers to access to abuse-deterrent opioid formulations by Medicare Part C and D beneficiaries. It also includes a number of requirements directed at reducing the potential for oversupply of opioids to reduce the potential for misuse and diversion. |
Properties
Our principal executive office is located at 7946 Ivanhoe Ave., Suite 201 in La Jolla, California, where we lease a total of 850 square feet of office space that we use for our administrative activities. All development activities are undertaken at contract research organizations. The lease expires in October 2024. We believe that our current arrangements will be sufficient to meet our needs for the foreseeable future, and that, should it be needed, suitable space will be available to accommodate our administrative activities.
Human Capital Resources (Employees)
We have seven full-time employees, one part-time employee and one consultant. Of these, five have a Ph.D. From time to time, we also retain independent contractors to support our organization. None of our employees is represented by a labor union or covered by collective bargaining agreements, and we believe our relationship with our employees is good.
| 32 |
Item 1A. Risk Factors
Risks Related to Our Business, Financial Condition and Capital Requirements
There is substantial doubt about our ability to continue as a going concern.
The Company does not have revenue generating activities and is dependent on additional financing to fund operations. These conditions raise substantial doubt about the Company’s ability to continue as a going concern. While we believe that we will be able to raise the capital we need to continue our operations, there can be no assurances that we will be successful in these efforts or will be able to resolve our liquidity issues or eliminate our operating losses. If we are unable to obtain sufficient funding, we would need to significantly reduce our operating plans and curtail some or all of our product development activities. Accordingly, our business, prospects, financial condition and results of operations will be materially and adversely affected, and we may be unable to continue as a going concern. If we seek additional financing to fund our business activities in the future and there remains substantial doubt about our ability to continue as a going concern, investors or other financing sources may be unwilling to provide additional funding on commercially reasonable terms or at all.
We are a clinical-stage pharmaceutical company with a limited operating history. We have incurred significant financial losses since our inception and anticipate that we will continue to incur significant financial losses for the foreseeable future.
We are a clinical-stage pharmaceutical company with a limited operating history. We have not yet demonstrated an ability to generate revenues, obtain regulatory approvals, engage in clinical development beyond Phase 1 trials, manufacture any product on a commercial scale or arrange for a third party to do so on our behalf or enter into licensing arrangements to commercialize a product, or conduct sales and marketing activities necessary for successful product commercialization.
We have no products approved for commercial sale and we have not generated any revenue from product sales to date, nor do we expect to generate any significant revenue from product sales for the next few years. We will continue to incur significant research and development and other expenses related to our product development, preclinical and clinical activities and ongoing operations. As a result, we are not profitable and have incurred losses in each period since our inception. Net losses and negative cash flows have had, and will continue to have, an adverse effect on our stockholders’ equity and working capital. Net losses and negative cash flows have had, and will continue to have, an adverse effect on our stockholders’ equity and working capital. We expect to continue to incur significant losses for the foreseeable future as we continue our research and development of, and seek regulatory approvals for, our product candidates.
If we continue to suffer losses as we have since inception, investors may not receive any return on their investment and may lose their entire investment.
In addition, as a public company, we incur significant additional legal, accounting and other expenses that we did not incur as a private company as we:
| ● | meet the requirements and demands of being a public company; | |
| ● | expand our operational, financial and management systems and increase personnel to support our operations; | |
| ● | hire additional clinical, quality control, medical, scientific and other technical personnel to support our clinical operations; | |
| ● | advance our clinical-stage product candidate PF614 through clinical development; | |
| ● | advance our preclinical stage product candidates into clinical development; | |
| ● | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
| 33 |
| ● | undertake any pre-commercialization activities to establish sales, marketing and distribution capabilities for any product candidates for which we may receive regulatory approval in regions where we choose to commercialize our products on our own or jointly with third parties; | |
| ● | maintain, expand and protect our intellectual property portfolio; and | |
| ● | make milestone, royalty or other payments due under any future in-license or collaboration agreements. |
Pharmaceutical product development entails substantial upfront capital expenditures and significant risk that any potential product candidate will fail to demonstrate adequate efficacy or an acceptable safety profile, gain regulatory approval, secure market access and reimbursement and become commercially viable. Therefore, any investment in us would be highly speculative. Our prospects are subject to the costs, uncertainties, delays and difficulties frequently encountered by companies in clinical development, especially clinical-stage pharmaceutical companies such as ours. Any predictions you make about our future success or viability may not be as accurate as they would otherwise be if we had a longer operating history or a history of successfully developing and commercializing pharmaceutical products. We will likely encounter unforeseen expenses, difficulties, complications, delays and other known or unknown factors in achieving our business objectives.
Additionally, our expenses could increase beyond our expectations if we are required by the United States Food and Drug Administration, or FDA, or other regulatory authorities to perform clinical trials in addition to those that we currently expect to conduct, or if there are any delays in establishing appropriate manufacturing arrangements for or in completing our clinical trials or the development of any of our product candidates.
Our ability to generate revenue from any of our potential products is subject to our ability to obtain regulatory approval and fulfill numerous other requirements and we may never be successful in generating revenues or becoming profitable.
Our ability to become and remain profitable depends on our ability to generate revenue or execute other business development arrangements. We do not expect to generate significant revenue, if any, unless and until we are able to obtain regulatory approval for, and successfully commercialize the product candidates we are developing or may develop. Successful commercialization, to the extent it occurs, will require achievement of many key milestones, including demonstrating safety and efficacy in clinical trials, obtaining regulatory approval for these product candidates, manufacturing, marketing and selling, or entering into other agreements to commercialize, those products for which we may obtain regulatory approval, satisfying any post-marketing requirements and obtaining reimbursement for our products from private insurance or government payors. Because of the uncertainties and risks associated with these activities, we cannot accurately and precisely predict the timing and amount, if any, of revenues, the extent of any further losses or when we might achieve profitability. We may never succeed in these activities and, even if we do, we may never generate revenues that are sufficient enough for us to achieve profitability. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis.
Our failure to become and remain profitable may depress the market price of our common stock and could impair our ability to raise capital, expand our business, diversify our product offerings or continue our operations.
We require substantial additional funding. If we are unable raise capital when needed, we could be forced to delay, reduce or terminate our product discovery and development programs or commercialization efforts.
We are a clinical stage pharmaceutical company that will need to raise additional capital to continue to operate as a going concern. Our quarterly operating results are likely to show continued losses in the future. Our operations have consumed substantial amounts of cash since inception. We expect to continue to spend substantial amounts to continue the clinical and preclinical development of our product candidates, including our planned Phase 2 program for nafamostat and planned clinical trials for PF614 and PF614-MPAR. We will need to raise additional capital to complete our currently planned clinical trials and any future clinical trials. Other unanticipated costs may arise during our development efforts. If we can obtain marketing approval for product candidates that we develop, we would require significant additional amounts of funding to launch and commercialize such product candidates. We cannot reasonably estimate the actual amounts necessary to successfully complete the development and commercialization of any product candidate we develop and we will require substantial additional funding to complete the development and commercialization of our product candidates.
| 34 |
Our future need for additional funding depends on many factors, including:
| ● | the scope, progress, results and costs of researching and developing our current product candidates, as well as other additional product candidates we may develop and pursue in the future, including the costs related to preclinical and clinical development of the product; | |
| ● | the timing of, and the costs involved in, obtaining marketing approvals for our product candidates and any other additional product candidates we may develop and pursue in the future; |
| ● | the number of future product candidates that we may pursue and their development requirements; | |
| ● | subject to receipt of regulatory approval, the costs of commercialization activities for our product candidates, to the extent such costs are not the responsibility of any future collaborators, including the costs and timing of establishing product sales, marketing, distribution and manufacturing capabilities; | |
| ● | subject to receipt of regulatory approval, the amount of revenue, if any, received from commercial sales of our product candidates or any other additional product candidates we may develop and pursue in the future; | |
| ● | the extent to which we in-license or acquire rights to other products, product candidates or technologies; | |
| ● | our ability to establish collaboration arrangements for the development of our product candidates on favorable terms, if at all; | |
| ● | dependent on financing, our headcount growth and associated costs as we expand our research and development and establishes a commercial infrastructure; | |
| ● | the costs of preparing, filing and prosecuting patent applications, maintaining and protecting our intellectual property rights, including enforcing and defending intellectual property related claims; and | |
| ● | the costs of operating as a public company. |
A change in the outcome of any of these or other factors with respect to the development of any of our product candidates could significantly change the costs and timing associated with the development of that product candidate, and many of these factors are outside of our control. Identifying potential product candidates and conducting preclinical studies and clinical trials is a time-consuming, expensive and uncertain process that takes years to complete, and we may never generate the necessary data or results required to obtain regulatory and marketing approval and achieve product sales. In addition, our product candidates, if approved, may not achieve commercial success. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. We cannot be certain that additional funding will be available on acceptable terms, or at all. Please see the risk factors under “Risks Related to the Ownership of Common Stock and Financial Reporting.”
We believe that our existing cash and cash equivalents will enable us to fund our operating expenses and capital expenditure requirements into the third quarter of 2024, while advancing our main product candidates such as, PF614 and PF614-MPAR and nafamostat through their respective next phases of clinical development. Our estimate may prove to be wrong, and we could use our available capital resources, if any, sooner than we currently expect. Further, changing circumstances, some of which may be beyond our control, could cause us to consume capital significantly faster than we currently anticipate, and we may need to seek additional funds sooner than planned. To the extent this occurs, it could impose significant dilution on our stockholders.
We may seek additional capital due to favorable market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. Attempting to secure additional financing may divert our management from our day-to-day activities, which may adversely affect our ability to develop our product candidates. Our failure to raise capital as and when needed or on acceptable terms would have a negative impact on our financial condition and our ability to pursue our business strategy, and we may have to delay, reduce the scope of, suspend or eliminate one or more of our platforms, programs, planned clinical trials or future commercialization efforts.
| 35 |
There may be no proceeds under the GEM Agreement or proceeds may be less than anticipated. The issuances of common stock pursuant to the GEM Agreement would result in dilution of existing stockholders and could have a negative impact on the market price of our common stock. Additionally, the negative covenants under the GEM Agreement are onerous and any breach by us thereunder may entitle GEM Global and GYBL to indemnification payments, reimbursements of legal and other expenses and other compensation thereby diverting our time and resources.
While we have raised capital from other sources, we have not used the GEM Facility to date. Under a Share Purchase Agreement between us, GEM Global Yield LLC SCS (“GEM Global”) and GEM Yield Bahamas Limited (“GYBL”), dated as of December 29, 2020, including a Registration Rights Agreement between the same parties and dated as of the same date (the “GEM Agreement”), we are entitled to draw down up to $60 million of gross proceeds from GEM Global in exchange for shares of our common stock at a price equal to 90% of the average closing bid price of the shares of our common stock on Nasdaq for a 30 day period, subject to meeting the terms and conditions of the GEM Agreement. This equity line facility is available for a period of 36 months from the closing date of the Merger. However, we have not been able to make use of the GEM Facility and we may not be able to do so before it expires. Please see the section entitled “Item1. Business” for additional information. The limitations on the amount and frequency of the draws that we can make pursuant to the GEM Agreement, which include the requirement that (i) there be an effective registration statement and (ii) size restrictions relating to our trading volume, may affect the ability to draw under the GEM Agreement and result in proceeds that are less than anticipated.
In addition, the occurrence of the Merger triggered (i) payment of a commitment fee of $1.2 million to GEM Global payable in either our common stock or cash, of which all has been satisfied with 3,838 shares of common stock transferred from related parties in July 2022 and an additional 44,444 shares of common stock issued in January 2023 and (ii) the issuance of a warrant granting GYBL the right to purchase 4,608 shares of our common stock, at a strike price per share reset to $1.5675. The number of shares underlying the warrant as well as the strike price is subject to adjustments for recapitalizations, reorganizations, change of control, stock split, stock dividend, reverse stock splits and certain issuances of additional shares of our common stock.
The issuances of shares at discount under the GEM Agreement and the anti-dilution protection granted to GEM Global in connection with issuances of additional shares of our common stock, would result in dilution of existing stockholders and have a negative impact on the market price of our common stock and our ability to obtain equity financing.
In addition, the negative covenants under the GEM Agreement are onerous and any breach thereof may trigger indemnification, reimbursement of losses and other liability for us thereby diverting our time and resources. To date, we have not used the GEM facility to raise capital.
Our business is highly dependent on the success of our product candidates. If we are unable to successfully complete clinical development, obtain regulatory approval for or commercialize one or more of our product candidates, or if we experience delays in doing so, our business will be materially harmed.
Our future success and ability to generate significant revenue from our product candidates, which we do not expect will occur for several years, is dependent on our ability to successfully develop, obtain regulatory approval for and commercialize one or more of our product candidates. A Phase 1b study of PF614 was initiated in 2021. Part A of the study completed enrollment in December 2021 and Part B was completed mid-year 2022. Two Human Abuse Potential clinical studies were completed in 2023. An efficacy Phase 2 study of PF614 was completed in December 2023. A Phase 1 trial was also initiated for PF614-MPAR in December 2021 and the clinical portion of Part A of that trial was completed in December 2022. The clinical portion of Part B was initiated in January 2023 and enrollment completed in March 2023. A Phase 1 safety study of nafamostat was completed in 2020. All of our other product candidates are in earlier stages of development and will require substantial additional investment for manufacturing, preclinical testing, clinical development, regulatory review and approval in one or more jurisdictions. If any of our product candidates encounter safety or efficacy problems, development delays or regulatory issues or other problems, our development plans and business would be materially harmed.
| 36 |
We may not have the financial resources to continue development of our product candidates. Even if clinical trials are completed, we may experience other issues that may delay or prevent regulatory approval of, or our ability to commercialize, our product candidates, including:
| ● | inability to demonstrate to the satisfaction of the FDA or comparable foreign regulatory authorities that our product candidates are safe and effective; | |
| ● | insufficiency of our financial and other resources to complete the necessary clinical trials and preclinical studies; |
| ● | negative or inconclusive results from our clinical trials, preclinical studies or the clinical trials of others for product candidates that are similar to ours, leading to a decision or requirement to conduct additional clinical trials or preclinical studies or abandon a program; | |
| ● | product-related adverse events experienced by subjects in our clinical trials, including unexpected toxicity results, or by individuals using drugs or therapeutic biologics similar to our product candidates; | |
| ● | delays in submitting an Investigational New Drug application, or IND, or comparable foreign applications or delays or failure in obtaining the necessary approvals from regulators to commence a clinical trial or a suspension or termination, or hold, of a clinical trial once commenced; | |
| ● | conditions imposed by the FDA, the European Medicines Agency, or EMA, or comparable foreign regulatory authorities regarding the scope or design of our clinical trials; | |
| ● | poor effectiveness of our product candidates during clinical trials; | |
| ● | better than expected performance of control arms, such as placebo groups, which could lead to negative or inconclusive results from our clinical trials; | |
| ● | delays in enrolling subjects in clinical trials; | |
| ● | high drop-out rates of subjects from clinical trials; | |
| ● | inadequate supply or quality of product candidates or other materials necessary for the conduct of our clinical trials; | |
| ● | greater than anticipated clinical trial or manufacturing costs; |
| ● | unfavorable FDA, EMA or comparable regulatory authority inspection and review of a clinical trial site; | |
| ● | failure of our third-party contractors or investigators to comply with regulatory requirements or the clinical trial protocol or otherwise meet their contractual obligations in a timely manner, or at all; | |
| ● | unfavorable FDA, EMA or comparable regulatory authority inspection and review of manufacturing facilities or inability of those facilities to maintain a compliance status acceptable to the FDA, EMA or comparable regulatory authorities; | |
| ● | delays and changes in regulatory requirements, policy and guidelines, including the imposition of additional regulatory oversight around clinical testing generally or with respect to our therapies in particular; or | |
| ● | varying interpretations of data by the FDA, EMA and comparable foreign regulatory authorities. |
| 37 |
Our product candidates will require additional, time-consuming development efforts prior to commercial sale, including preclinical studies, clinical trials and approval by the FDA and applicable foreign regulatory authorities. All product candidates are prone to the risks of failure that are inherent in pharmaceutical product development, including the possibility that such product candidate will not be shown to be sufficiently safe and effective for approval by regulatory authorities. In addition, we cannot assure stockholders that any such products that are approved will be manufactured or produced economically, successfully commercialized or widely accepted in the marketplace or be more effective than other commercially available alternatives.
We depend heavily on the success of our lead product candidate PF614, which is currently in clinical trials. Our clinical trials of PF614 may not be successful. If we are unable to commercialize PF614 or experience significant delays in doing so, our business will be materially harmed.
We have invested a significant portion of our efforts and financial resources in the research and development of our lead product candidate, PF614 and we expect to continue to do so. Our ability to generate revenues from the sale of abuse-deterrent opioid products, which may not occur at a significant level for several years, will depend heavily on the successful development, regulatory approval and eventual commercialization of PF614.
We cannot commercialize product candidates in the United States without first obtaining regulatory approval for the product from the FDA; similarly, we cannot commercialize product candidates outside of the United States without obtaining regulatory approval from similar regulatory authorities outside of the United States. Even if PF614 or another product candidate were to successfully obtain approval from the FDA and non-U.S. regulatory authorities, any approval might contain significant limitations related to use restrictions for specified age groups, warnings, precautions or contraindications, or may be subject to burdensome post-approval study or risk management requirements. If we are unable to obtain regulatory approval for PF614 in one or more jurisdictions, or any approval contains significant limitations, we may not be able to obtain sufficient funding or generate sufficient revenue to continue the development, marketing and/or commercialization of PF614 or any other product candidate that we may discover, in-license, develop or acquire in the future. Furthermore, even if we obtain regulatory approval for PF614, we will still need to develop a commercial organization, or collaborate with third parties for the commercialization of PF614, establish commercially viable pricing and obtain approval for adequate reimbursement from third-party and government payors. If we or our commercialization collaborators are unable to successfully commercialize PF614, we may not be able to generate sufficient revenues to continue our business.
Due to the significant resources required for the development of our product pipeline, and depending on our ability to access capital, we must prioritize the development of certain product candidates over others. Moreover, we may fail to expend our limited resources on product candidates or indications that may have been more profitable or for which there is a greater likelihood of success.
We currently have three clinical-stage product candidates as well as certain other product candidates that are at various stages of preclinical development. We seek to maintain a process of prioritization and resource allocation to maintain an optimal balance between aggressively pursuing our more advanced clinical-stage product candidates, such as PF614 and PF614-MPAR, and ensuring the development of additional potential product candidates.
Due to the significant resources required for the development of our product candidates, we must focus on specific diseases and disease pathways and decide which product candidates to pursue and advance and the amount of resources to allocate to each. Our decisions concerning the allocation of research, development, collaboration, management and financial resources toward particular product candidates or therapeutic areas may not lead to the development of any viable commercial products and may divert resources away from better opportunities. If we make incorrect determinations regarding the viability or market potential of any of our product candidates or misinterpret trends in the pharmaceutical industry, in particular for opioid abuse and drug overdose, our business, financial condition, and results of operations could be materially adversely affected. As a result, we may (i) fail to capitalize on viable commercial products or profitable market opportunities, (ii) be required to forego or delay pursuit of opportunities with other product candidates or other diseases and disease pathways that may later prove to have greater commercial potential than those we choose to pursue, or (iii) relinquish valuable rights to such product candidates through collaboration, licensing, or other royalty arrangements in cases in which it would have been advantageous for us to invest additional resources to retain sole development and commercialization rights.
| 38 |
Our PF614 and PF614-MPAR product candidates may not be successful in limiting or impeding abuse, overdose or misuse or providing additional safety upon commercialization.
We are committing a substantial majority of our resources to the development of products utilizing our TAAP and MPAR®. There can be no assurance that our products will perform as tested and limit or impede the actual abuse, overdose or misuse of such products or provide other benefits in commercial settings. Moreover, there can be no assurance that if our products are approved by the FDA, the post-approval epidemiological studies required by the FDA as a condition of any such approvals of the products will show a reduction in the consequences of abuse and misuse by patients for whom the applicable product is prescribed. The failure of our products to limit or impede actual abuse, overdose or misuse or provide other safety benefits in practice will have a material adverse impact on market acceptance for such products and on our financial condition and results of operations.
If we do not achieve our projected development and commercialization goals within the timeframes we expect, the development and commercialization of our product candidates may be delayed, and our business and results of operations may be harmed.
For planning purposes, we seek to estimate the timing of the accomplishment of various scientific, clinical, regulatory and other product development objectives. These milestones may include our expectations regarding the commencement or completion of scientific studies and clinical trials, the submission of regulatory filings, or commercialization objectives. From time to time, we may publicly announce the expected timing of some of these milestones, such as the completion of an ongoing clinical trial, the initiation of other clinical programs, receipt of marketing approval or a commercial launch of a product. The potential achievement of many of these milestones may be outside of our control. Each of these milestones is based on a variety of assumptions which, if not realized as expected, may cause the timing of such potential achievement of the respective milestones to vary considerably from our estimates, including:
| ● | our available capital resources or capital constraints we experience; | |
| ● | the rate of progress, costs and results of our clinical trials and research and development activities, including the extent of scheduling conflicts with participating clinicians and collaborators; | |
| ● | our ability to identify and enroll patients who meet clinical trial eligibility criteria; | |
| ● | our receipt of approvals by the FDA and other regulatory authorities and the timing thereof; | |
| ● | clinical outcomes; | |
| ● | other actions, decisions or rules issued by regulators; | |
| ● | our ability to access sufficient, reliable and affordable supplies of materials used in the manufacture of our product candidates; | |
| ● | the efforts of our collaborators with respect to the commercialization of our product candidates; and | |
| ● | the securing of, costs related to, and timing issues associated with, product manufacturing as well as sales and marketing activities. |
If we fail to achieve any announced milestones in the timeframes we expect, the development and commercialization of our product candidates may be delayed, and our business and results of operations may be harmed and it could negatively impact our share price performance. Please see “Business” for more information.
| 39 |
Competitive products may reduce or eliminate commercial opportunity for our product candidates, if approved. If our competitors develop technologies or product candidates more rapidly than we do, or their technologies or product candidates are more effective or safer than any such technologies or product candidate of ours, our ability to develop and successfully commercialize our own technologies or product candidates may be adversely affected.
The clinical and commercial landscapes for the solution of opioid abuse and drug overdose are highly competitive and subject to rapid and significant technological change. We face competition with respect to our indications for our product candidates and will face competition with respect to any other product candidates that we may seek to develop or commercialize in the future, from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. There are several large pharmaceutical and biotechnology companies that currently market and sell drugs or are pursuing the development of product candidates for the treatment of the indications that we are pursuing. These companies include, but are not limited to, Purdue Pharma, LP, and Collegium Pharmaceutical, Inc. Potential competitors include companies developing novel non-opioid pain drug candidates such as pharmaceutical companies and academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
We believe that a significant number of product candidates are currently under development for the same indications that we are currently pursuing, and some or all may become commercially available in the future for the treatment of conditions for which we are trying or may try to develop product candidates. Our potential competitors include large pharmaceutical and biotechnology companies, specialty pharmaceutical and generic drug companies, academic institutions, government agencies and research institutions. See the section entitled “Business — Competition” for examples of the competition that our product candidates face.
Our competitors may have significantly greater financial resources, established presence in the market, expertise in research and development, manufacturing, preclinical and clinical testing, obtaining regulatory approvals and reimbursement and marketing approved products than us. Accordingly, our competitors may be more successful than we may be in obtaining regulatory approval for therapies and achieving widespread market acceptance. Our competitors’ products may be more effective, or more effectively marketed and sold, than any product candidate we may commercialize and may render our therapies obsolete or non-competitive before we can recover development and commercialization expenses. If any of our product candidates, including PF614, is approved, these product candidates could compete with a range of therapeutic treatments that are in development. In addition, our competitors may succeed in developing, acquiring or licensing technologies and products that are more effective or less costly than PF614, our other product candidates or any other product candidates that we may develop, which could render our product candidates obsolete and noncompetitive.
If we obtain approval for any of our product candidates, we may face competition based on many different factors, including the efficacy, safety and tolerability of our products, the ease with which our products can be administered, the timing and scope of regulatory approvals for these products, the availability and cost of manufacturing, marketing and sales capabilities, price, reimbursement coverage and patent position. Existing and future competing products could present superior treatment alternatives, including being more effective, safer, less expensive or marketed and sold more effectively than any products we may develop.
Competitive products may make any products we develop obsolete or noncompetitive before we are able to recover the expense of developing and commercializing our product candidates. Such competitors could also recruit our employees, which could negatively impact our level of expertise and our ability to execute our business plan.
In addition, our competitors may obtain patent protection, regulatory exclusivities or FDA approval and commercialize products more rapidly than we do, if we are successful at all, which may impact future approvals or sales of any of our product candidates that receive regulatory approval. If the FDA approves the commercial sale of PF614 or any other product candidate, we will also be competing with respect to marketing capabilities and manufacturing efficiency. We expect any such competition among products will be based on product efficacy and safety, the timing and scope of regulatory approvals, availability of supply, marketing and sales capabilities, product price, reimbursement coverage by government and private third-party payors, regulatory exclusivities and patent position. Our profitability and financial position will suffer if our product candidates receive regulatory approval but cannot compete effectively in the marketplace.
Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites, as well as in acquiring technologies complementary to, or necessary for, our programs.
| 40 |
Our business could be harmed if we lose the services of our key personnel or if we are unable to hire additional highly qualified employees.
Our business depends upon our ability to attract and retain highly qualified personnel, including managerial, sales and technical personnel. We compete for key personnel with other companies, healthcare institutions, academic institutions, government entities and other organizations. Our ability to maintain and expand our business may be impaired if we are unable to retain our current key personnel or hire or retain other qualified personnel in the future.
We currently only have seven full-time employees, one part-time employee and one consultant and we expect to add additional employees. Our future success also depends on our ability to identify, attract, hire or engage, retain and motivate other well-qualified managerial, technical, clinical and regulatory personnel.
Competition for such individuals, particularly in the United States, is intense, and we may not be able to hire sufficient personnel to support our efforts. There can be no assurance that such professionals will be available in the market, or that we will be able to retain existing professionals or to meet or to continue to meet their compensation requirements. Furthermore, our cost base with respect to such compensation, which may include equity compensation, may increase significantly, which could have a material adverse effect on our financial results, including the potential for additional dilution to our stockholders. Failure to establish and maintain an effective management team and work force could adversely affect our ability to operate, grow and manage our business.
Our employees, independent contractors, principal investigators, consultants, commercial collaborators, service providers and other vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have an adverse effect on our results of operations.
We are exposed to the risk that we and our contract research organizations’ (“CROs”) employees and contractors, including principal investigators, consultants, commercial collaborators, service providers and other vendors may engage in fraudulent or other illegal activity. Misconduct by these parties could include intentional, reckless and/or negligent conduct or other unauthorized activities that violate the laws and regulations of the FDA and other similar regulatory bodies, including those laws that require the reporting of true, complete and accurate information to such regulatory bodies; manufacturing standards; federal and state healthcare fraud and abuse and health regulatory laws and other similar foreign fraudulent misconduct laws; or laws that require the true, complete and accurate reporting of financial information or data. Activities subject to these laws also involve the improper use or misrepresentation of information obtained during clinical trials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter third-party misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and financial results, including the imposition of significant civil, criminal and administrative penalties, damages, monetary fines, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations
Some of our programs are partially supported by government grant awards, which may not be available to us in the future.
We have received funding under grant award programs funded by governmental agencies, such as the NIH and NIDA. To fund a portion of our future research and development programs, we may apply for additional grant funding from these or similar governmental agencies in the future. However, funding by these, and other, governmental agencies may be significantly reduced or eliminated in the future for several reasons. For example, some programs are subject to a yearly appropriations process in Congress. In addition, we may not receive full funding under current or future grants because of budgeting constraints of the agency administering the program or unsatisfactory progress on the study being funded. Therefore, we cannot provide any assurance that we will receive any future grant funding from any government agencies, or, if received, we will receive the full amount of the particular grant award. Any such reductions could delay the development of our product candidates and the introduction of new products.
| 41 |
Risks Related to Our Dependence on Third-Party Providers
We currently rely on, and expect to rely on in the future, third parties to conduct our clinical trials, and those third parties may not perform satisfactorily, including failing to meet deadlines for completing such trials, failing to satisfy legal or regulatory requirements or terminating the relationship.
We currently rely on, and expect to rely on in the future, third-party CROs to conduct research and development activities and our clinical trials for our product candidates. Agreements with these CROs might terminate for a variety of reasons, including for their failure to perform. Entry into alternative arrangements, if necessary, could significantly delay our product development activities.
Our reliance on these CROs for research and development activities and clinical trials will reduce our control over these activities but will not relieve us of any of our responsibilities. For example, we will remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols in the applicable IND. Moreover, the FDA requires compliance with standards, commonly referred to as good clinical practices, or GCPs, for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected.
If these CROs do not successfully carry out their contractual duties, meet expected deadlines or conduct the clinical trials in accordance with regulatory requirements or our stated protocols, it could adversely affect the development of our product candidates and it could result in us not being able to obtain, or being delayed in obtaining, marketing approvals for our product candidates and it could adversely affect our efforts to successfully commercialize our product candidates.
We expect to be completely dependent on third parties to manufacture our product candidates, and our commercialization of our product candidates could be halted, delayed or made less profitable if those third parties fail to maintain a compliance status acceptable to the FDA or comparable foreign regulatory authorities, fail to provide to us with sufficient quantities of our product candidates or fail to do so at acceptable quality levels or prices.
We do not currently have, nor do we plan to acquire, the capability or infrastructure to manufacture the ingredients in our product candidates for use in our clinical trials or for commercial product, if any. We have entered into an agreement with Purisys LLC (“Purisys”) for production of PF614 drug substance and a Manufacturing Agreement (the “Recro Agreement”) with Recro Gainesville LLC (“Recro”) now known as Societal CDMO, (“Societal”) for the production of PF614 capsules and other materials and services with respect to our clinical studies. In addition, we do not have the capability to encapsulate any of our product candidates as a finished product for commercial distribution. As a result, we expect to be obligated to rely on contract manufacturers, like Societal, if and when any of our product candidates are approved for commercialization. In the event that Societal is unable to perform its obligations under the Recro Agreement, we may be unable to replace the Recro Agreement on terms as favorable to us. We have not entered into an agreement with any contract manufacturers for commercial supply and may not be able to engage a contract manufacturer for commercial supply of any of our product candidates on favorable terms to us, or at all.
The processes used by our contract manufacturers to manufacture our product candidates must be approved by the FDA or comparable foreign regulatory authorities and the facilities at which the product candidates are manufactured must maintain a compliance status acceptable to the FDA and foreign regulatory authorities. FDA and foreign regulatory authorities will conduct inspections after we submit a new drug application, or NDA, to the FDA or its equivalent to other relevant regulatory authorities. We will not control the manufacturing process of, and will be completely dependent on, its contract manufacturing partners for compliance with cGMPs for manufacture of both active drug substances and finished products. These cGMP regulations cover all aspects of the manufacturing, testing, quality control and record keeping relating to our product candidates. If our contract manufacturers, including Societal, do not successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA or others, our product candidates may not be approved. If these facilities do not maintain a compliance status acceptable to the FDA, Drug Enforcement Agency, or DEA, or comparable regulatory authorities, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, if approved.
| 42 |
Our contract manufacturers, including Purisys and Societal, will be subject to ongoing periodic unannounced inspections by the FDA, DEA and corresponding state and foreign agencies for compliance with cGMPs, security, recordkeeping and similar regulatory requirements. Although we will not have control over our contract manufacturers’ compliance with these regulations and standards, we are nonetheless responsible for assuring such compliance. Failure by any of our contract manufacturers to comply with applicable regulations could result in sanctions being imposed on us, including fines, injunctions, civil penalties, failure to grant approval to market any of our product candidates, delays, suspensions or withdrawals of approvals, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect our business and results of operations. Failure by our contract manufacturers to comply with or maintain any of these standards could adversely affect our ability to develop, obtain regulatory approval for or market any of our product candidates.
If, for any reason, these third parties, including Societal, are unable or unwilling to perform, we may not be able to terminate our agreements with them, and we may not be able to locate alternative manufacturers or formulators or enter into favorable agreements with them and we cannot be certain that any such third parties will have the manufacturing capacity to meet future requirements. If these manufacturers or any alternate manufacturer of finished drug product experiences any significant difficulties in its respective manufacturing processes for our ingredients or finished products or should cease doing business with us, we could experience significant interruptions in the supply of any of our product candidates or may not be able to create a supply of our product candidates at all. Our inability to coordinate the efforts of our third-party manufacturing partners, or the lack of capacity available at our third-party manufacturing partners, could impair our ability to supply any of our product candidates at required levels. Because of the significant regulatory requirements that we would need to satisfy in order to qualify a new bulk or finished product manufacturer, if we face these or other difficulties with our current manufacturing partners, we could experience significant interruptions in the supply of any of our product candidates if we decide to transfer the manufacture of any of our product candidates to one or more alternative manufacturers in an effort to deal with the difficulties.
Any manufacturing problem or the loss of a contract manufacturer, including Societal, could be disruptive to our operations and delay development of our investigational products. Additionally, we rely on third parties to supply the raw materials needed to manufacture our potential products. Any reliance on suppliers may involve several risks, including a potential inability to obtain critical materials and reduced control over production costs, delivery schedules, reliability and quality. Any unanticipated disruption to a future contract manufacturer caused by problems at suppliers could delay shipment of any of our investigational products and, if approved, product candidates.
We cannot guarantee that our future manufacturing and supply partners will be able to reduce the costs of commercial scale manufacturing of any of our product candidates over time. If the commercial-scale manufacturing costs of any of our product candidates are higher than expected, these costs may significantly impact our operating results. In order to reduce costs, we may need to develop and implement process improvements. However, in order to do so, we will need, from time to time, to notify or make submissions with regulatory authorities, and the improvements may be subject to approval by such regulatory authorities.
We cannot be sure that we will receive these necessary approvals or that these approvals will be granted in a timely fashion. We also cannot guarantee that we will be able to enhance and optimize output in our commercial manufacturing process. If we cannot enhance and optimize output, we may not be able to reduce our costs over time.
If we are unable to develop our sales, marketing and distribution capability on our own or through collaborations with marketing partners, we will not be successful in commercializing our product candidates.
We currently have no marketing, sales or distribution capabilities. We intend to establish a sales and marketing organization, either on our own or in collaboration with third parties, with technical expertise and supporting distribution capabilities to commercialize PF614 or one or more of our other product candidates that may receive regulatory approval in key territories. These efforts will require substantial additional resources, some or all of which may be incurred in advance of any approval of the product candidate. Any failure or delay in the development of our or third parties’ internal sales, marketing and distribution capabilities would adversely impact the commercialization of PF614, our other product candidates and other future product candidates.
| 43 |
Factors that may inhibit our efforts to commercialize our product candidates on our own include:
| ● | our inability to recruit and retain effective sales and marketing personnel; | |
| ● | the inability of sales personnel to obtain access to or persuade physicians to prescribe any future products; | |
| ● | the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and | |
| ● | unforeseen costs and expenses associated with creating an independent sales and marketing organization. |
With respect to our existing and future product candidates, we may choose to collaborate with third parties that have direct sales forces and established distribution systems to serve as an alternative to our own sales force and distribution systems. Our future product revenue may be lower than if we directly marketed or sold our product candidates, if approved. In addition, any revenue we receive will depend in whole or in part upon the efforts of these third parties, which may not be successful and are generally not within our control. If we are not successful in commercializing any approved products, our future product revenue will suffer, and we may incur significant additional losses.
If we do not establish sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
Risks Related to Product Development, Regulatory Approval, Manufacturing and Commercialization
The regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time-consuming and inherently unpredictable, and if we are ultimately unable to obtain regulatory approval for our product candidates, our business will be substantially harmed.
We are not permitted to commercialize, market, promote or sell any product candidate in the United States without obtaining regulatory approval from the FDA. Foreign regulatory authorities, such as the EMA, impose similar requirements. The time required to obtain approval by the FDA and comparable foreign authorities is inherently unpredictable, but typically takes many years following the commencement of clinical trials and depends upon numerous factors, including substantial discretion of the regulatory authorities. In addition, approval policies, regulations, or the type and amount of clinical data necessary to gain approval may change during a product candidate’s clinical development and may vary among jurisdictions. To date, we have not submitted an NDA to the FDA or similar drug approval submissions to comparable foreign regulatory authorities for our most advanced product candidate, PF614, or any other product candidate. We must complete additional preclinical studies and clinical trials to demonstrate the safety and efficacy of our product candidates in humans before we will be able to obtain these approvals.
Clinical testing is expensive, difficult to design and implement, can take many years to complete and is inherently uncertain as to outcome. We cannot guarantee that any clinical trials will be conducted as planned or completed on schedule, if at all. The clinical development of our initial and potential additional product candidates is susceptible to the risk of failure inherent at any stage of development, including failure to demonstrate efficacy in a clinical trial or across a broad population of patients, the occurrence of adverse events that are severe or medically or commercially unacceptable, failure to comply with protocols or applicable regulatory requirements, and determination by the FDA or any comparable foreign regulatory authority that a product candidate may not continue development or is not approvable. It is possible that even if any of our product candidates has a beneficial effect, that effect will not be detected during clinical evaluation because of one or more of a variety of factors, including the size, duration, design, measurements, conduct or analysis of our clinical trials. Conversely, because of the same factors, our clinical trials may indicate an apparent positive effect of such product candidate that is greater than the actual positive effect, if any. Similarly, in our clinical trials, we may fail to detect toxicity of, or intolerability caused by, such product candidate, or mistakenly believe that our product candidates are toxic or not well tolerated when that is not in fact the case. Serious adverse events, or SAEs, or other adverse effects, as well as tolerability issues, could hinder or prevent market acceptance of the product candidate at issue.
| 44 |
Our current and future product candidates could fail to receive regulatory approval for many reasons, including the following:
| ● | the FDA or comparable foreign regulatory authorities may disagree as to the design or implementation of our clinical trials; | |
| ● | we may be unable to demonstrate to the satisfaction of the FDA or comparable foreign regulatory authorities that a product candidate is safe and effective for our proposed indication; | |
| ● | the results of clinical trials may not meet the level of statistical significance required by the FDA or comparable foreign regulatory authorities for approval; | |
| ● | we may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks; |
| ● | the FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from clinical trials or preclinical studies; | |
| ● | the data collected from clinical trials of our product candidates may not be sufficient to support the submission of an NDA to the FDA or other submission or to obtain regulatory approval in the United States, the European Union or elsewhere; | |
| ● | the FDA or comparable foreign regulatory authorities may find deficiencies with the manufacturing processes of third-party manufacturers with which we contract for clinical and commercial supplies; and | |
| ● | the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval. |
This lengthy approval process as well as the unpredictability of clinical trial results may result in us failing to obtain regulatory approval to market any product candidate we develop, which would substantially harm our business, results of operations and prospects. The FDA and other comparable foreign authorities have substantial discretion in the approval process and determining when or whether regulatory approval will be granted for any product candidate that we develop. Even if we believe the data collected from future clinical trials of our product candidates are promising, such data may not be sufficient to support approval by the FDA or any other regulatory authority.
In addition, even if we were to obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited indications than we request, may not approve the price we intend to charge for our products, may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve a product candidate with labeling that does not include the claims necessary or desirable for the successful commercialization of that product candidate. Any of the foregoing scenarios could materially harm the commercial prospects for our product candidates.
The FDA may recommend scheduling with respect to any of our current or future product candidates. In such event, prior to a product launch, the DEA will need to determine the controlled substance schedule of the product, considering the recommendation of the FDA. The timing of the scheduling process is uncertain and may delay our ability to market any product candidate that we successfully developed and approved.
| 45 |
If our clinical trials fail to replicate positive results from earlier preclinical studies or clinical trials conducted by us or third parties, we may be unable to successfully develop, obtain regulatory approval for, or commercialize our product candidates.
The results observed from preclinical studies or early-stage clinical trials of our product candidates may not necessarily be predictive of the results of later-stage clinical trials that we conduct. Similarly, positive results from such preclinical studies or early-stage clinical trials may not be replicated in our subsequent preclinical studies or clinical trials. For example, preclinical studies showed that PF614 does not readily convert into oxycodone in the blood stream following direct injection, and the Phase 1 trial we have conducted with TAAP prodrug PF614 (“prodrug”: a medication or compound that, after administration, is metabolized, i.e., converted within the body into a pharmacologically active drug), demonstrated that, after oral administration, the corresponding opioid was measured in the subjects’ blood. Furthermore, our product candidates may not be able to demonstrate similar activity or adverse event profiles as other product candidates that we believe may have similar profiles.
There can be no assurance that any of our clinical trials will ultimately be successful or support further clinical development of any of our product candidates. There is a high failure rate for drugs proceeding through clinical trials. Many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in late-stage clinical trials after achieving positive results in early-stage development, and we cannot be certain that we will not face similar setbacks. These setbacks have been caused by, among other things, preclinical findings made while clinical trials were underway or safety or efficacy observations made in preclinical studies and clinical trials, including previously unreported adverse events.
Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials nonetheless failed to obtain FDA, EMA or comparable foreign regulatory authority approval.
The FDA, EMA or comparable foreign regulatory authorities may disagree with our regulatory plan for our product candidates.
We hold INDs for PF614 and nafamostat and completed a Phase 1 trial for each product candidate. We have completed multiple human abuse potential and efficacy studies for PF614. We have applied for and received fast track designation for PF614. However, fast track designation does not guarantee a faster development or regulatory review or approval process and does not assure FDA approval. We have received feedback from the FDA on requirements to achieve abuse deterrent labeling claims for PF614. We have also received an IND for PF614-MPAR and have completed an initial Phase 1 study. We have been granted Breakthrough Therapy designation by the FDA for PF614-MPAR. For all INDs that we hold we have received feedback on required pre-clinical, manufacturing and clinical studies that will be required for an NDA.
Our clinical trial results may not support approval of our product candidates. The general approach for FDA approval of a new drug is dispositive data from two or more well-controlled Phase 3 clinical trials of the product candidate in the relevant patient population. Phase 3 clinical trials typically involve a large number of patients, have significant costs, and take years to complete. In addition, there is no assurance that the endpoints and trial designs that we intend to use for our planned clinical trials, including those that we have developed based on feedback from regulatory agencies or those that have been used for the approval of similar drugs, will be acceptable for future approvals.
Interim topline and preliminary data from our clinical trials that we announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publish interim topline or preliminary data from our clinical trials. Interim data from clinical trials that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Preliminary or topline data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, interim and preliminary data should be viewed with caution until the final data is available. Adverse differences between preliminary or interim data and final data could significantly harm our reputation and business prospects.
| 46 |
Even if we complete the necessary preclinical studies and clinical trials, the marketing approval process is expensive, time-consuming and uncertain and may prevent us from obtaining approvals for the commercialization of our product candidates.
Any product candidate we develop and the activities associated with such development and commercialization, including our design, testing, manufacture, safety, efficacy, recordkeeping, labeling, storage, approval, advertising, promotion, sale, and distribution, are subject to comprehensive regulation by the FDA and other regulatory authorities in the United States and by comparable authorities in other countries. Failure to obtain marketing approval for a product candidate will prevent us from commercializing the product candidate in a given jurisdiction. We have not received approval to market any product candidates from regulatory authorities in any jurisdiction and it is possible that none of the product candidates we are developing or may seek to develop in the future will ever obtain regulatory approval. Ensysce has no experience in submitting and supporting the applications necessary to gain marketing approvals and we expect to rely on third-party CROs or regulatory consultants to assist us in this process. Securing regulatory approval requires the submission of extensive preclinical and clinical data and supporting information to the various regulatory authorities for each therapeutic indication to establish the product candidate’s safety and efficacy. Securing regulatory approval also requires the submission of information about the product manufacturing process to, and inspection of manufacturing facilities by, the relevant regulatory authority. Any product candidates we develop may not be effective, may be only moderately effective, or may prove to have undesirable or unintended side effects, toxicities or other characteristics that may preclude us from obtaining marketing approval or prevent or limit commercial use.
The process of obtaining marketing approvals, both in the United States and abroad, is expensive, may take many years if additional clinical trials are required, if approval is obtained at all, and can vary substantially based upon a variety of factors, including the type, complexity, and novelty of the product candidates involved. Changes in marketing approval policies during the development period, changes in or the enactment of additional statutes or regulations, or changes in regulatory review for each submitted product application, may cause delays in the approval or rejection of an application. The FDA and comparable authorities in other countries have substantial discretion in the approval process and may refuse to accept any application or may decide that our data is insufficient for approval and requires additional preclinical, clinical or other studies. In addition, varying interpretations of the data obtained from preclinical and clinical testing could delay, limit, or prevent marketing approval of a product candidate. Any marketing approval that we may ultimately obtain could be limited or subject to restrictions or post-approval commitments that render the approved product not commercially viable. For example, during the product approval process, the FDA will determine whether a REMS plan is necessary to assure the safe use of the product. All opioid analgesic products currently on the market in the United States are subject to a REMS. A REMS may be required to include various elements, such as a medication guide or patient package insert, a communication plan to educate health care providers of the risks, limitations on who may prescribe or dispense the drug or other measures that the FDA deems necessary to assure the safe use of the drug. In addition, the REMS plan must include a timetable to assess the strategy at eighteen months, three years and seven years after approval. We may be required to develop a REMS for the product, or participate in a REMS with other manufacturers, or to develop a similar strategy as required by a regulatory authority.
Even if approved, our contract manufacturers will need to obtain quota from DEA to manufacture sufficient quantities and maintain inventories of product to be commercially distributed.
If we experience delays in obtaining manufacturing approval or if we fail to obtain manufacturing approval of any product candidates we may develop, the commercial prospects for those product candidates may be harmed, and our ability to generate revenues will be materially impaired.
| 47 |
We may incur unexpected costs or experience delays in completing, or ultimately be unable to complete, the preclinical and clinical studies necessary for development and commercialization of our product candidates.
To obtain the requisite regulatory approvals to commercialize any of our product candidates, we must demonstrate through extensive preclinical studies and clinical trials that our product candidates are safe and effective in humans. We may experience delays in completing our clinical trials or preclinical studies and initiating or completing additional clinical trials or preclinical studies, including as a result of regulators not allowing or delay in allowing clinical trials to proceed under an IND, or not approving or delaying approval for any clinical trial grant or similar approval that we need to initiate a clinical trial. We may also experience numerous unforeseen events during our clinical trials that could delay or prevent our ability to receive marketing approval or commercialize the product candidates we develop, including:
| ● | regulators, or institutional review boards, or IRBs, or other reviewing bodies may not authorize us or our investigators to commence a clinical trial, or to conduct or continue a clinical trial at a prospective or specific trial site; | |
| ● | we may not reach agreement on acceptable terms with prospective CROs and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; | |
| ● | we may experience challenges or delays in recruiting principal investigators or study sites to lead our clinical trials; | |
| ● | the number of subjects or patients required for clinical trials of our product candidates may be larger than we anticipate, enrollment in these clinical trials may be insufficient or slower than we anticipate, and the number of clinical trials being conducted at any given time may be high and result in fewer available patients for any given clinical trial, or patients may drop out of these clinical trials at a higher rate than we anticipate; | |
| ● | our third-party contractors, including those manufacturing our product candidates or conducting clinical trials on our behalf, may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, or at all; | |
| ● | we may have to amend clinical trial protocols submitted to regulatory authorities or conduct additional studies to reflect changes in regulatory requirements or guidance, which we may be required to resubmit to an IRB and regulatory authorities for re-examination; | |
| ● | regulators or other reviewing bodies may find deficiencies with or subsequently find fault with the manufacturing processes or facilities of third-party manufacturers with which we enter into agreement for clinical and commercial supplies, or the supply or quality of any product candidate or other materials necessary to conduct clinical trials of our product candidates may be insufficient, inadequate or not available at an acceptable cost, or we may experience interruptions in supply; and | |
| ● | the potential for approval policies or regulations of the FDA or the applicable foreign regulatory agencies to significantly change in a manner rendering our clinical data insufficient for approval. |
Regulators or IRBs of the institutions in which clinical trials are being conducted may suspend, limit or terminate a clinical trial, or data monitoring committees may recommend that we suspend or terminate a clinical trial, due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial operations or trial site by the FDA or other regulatory authorities resulting in the imposition of a clinical hold (such as the clinical hold placed on PF614-MPAR in January 2021), safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative actions, or lack of adequate funding to continue the clinical trial. Negative or inconclusive results from our clinical trials or preclinical studies could mandate repeated or additional clinical trials and, to the extent we choose to conduct clinical trials in other indications, could result in changes to or delays in clinical trials of our product candidates in such other indications. We do not know whether any clinical trials that we conduct will demonstrate adequate efficacy and safety to result in regulatory approval to market our product candidates for the indications that we are pursuing. If later-stage clinical trials do not produce favorable results, our ability to obtain regulatory approval for our product candidates will be adversely impacted.
| 48 |
Our failure to successfully initiate and complete clinical trials and to demonstrate the efficacy and safety necessary to obtain regulatory approval to market our product candidates would significantly harm its business. The development costs of our product candidates will also increase if we experience delays in testing or regulatory approvals and we may be required to obtain additional funds to complete clinical trials. We cannot assure stockholders that our clinical trials will begin as planned or be completed on schedule, if at all, or that we will not need to restructure or otherwise modify our trials after they have begun. Significant clinical trial delays could also shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before we do and impair our ability to successfully commercialize our product candidates, which may harm our business and results of operations. In addition, many of the factors that cause, or lead to, delays of clinical trials may ultimately lead to the denial of regulatory approval of our product candidates.
If we encounter difficulties enrolling patients in our clinical trials, our clinical development activities could be delayed or otherwise adversely affected.
The timely completion of clinical trials in accordance with our protocols depends on, among other things, our ability to enroll a sufficient number of patients who remain in the study until its conclusion.
We may experience difficulties in patient enrollment in our clinical trials for a variety of factors, including:
| ● | the patient eligibility criteria defined in the protocol; | |
| ● | the size of the patient population required for analysis of the trial’s primary endpoints; | |
| ● | the proximity of patients to study sites; | |
| ● | the design of the trial; | |
| ● | our ability to recruit clinical trial investigators with the appropriate competencies and experience; | |
| ● | competing clinical trials and clinicians’ and patients’ perceptions as to the potential advantages and risks of the product candidate being studied in relation to other available therapies, including any new drugs that may be approved for the indications that we are investigating; | |
| ● | our ability to obtain and maintain patient consents; and | |
| ● | the risk that patients enrolled in clinical trials will drop out of the trials before completion. |
In addition, our clinical trials may compete with other clinical trials for product candidates that are in the same therapeutic areas as our product candidates, and this competition will reduce the number and types of patients available to us, because some patients who might have opted to enroll in our trials may instead opt to enroll in a trial being conducted by one of our competitors. Since the number of qualified clinical investigators is limited, we may conduct some of our clinical trials at the same clinical trial sites that some of our competitors use, which will reduce the number of patients who are available for our clinical trials in such clinical trial site. Furthermore, if significant adverse events or other side effects are observed in any of our clinical trials, we may have difficulty recruiting patients to our trials and patients may drop out of our trials.
Our inability to enroll enough patients for our clinical trials would result in significant delays or might require us to abandon one or more clinical trials or our development efforts altogether. Delays in patient enrollment may result in increased costs, negatively affect the timing or outcome of the planned clinical trials, delay the product candidate development and approval process and jeopardize our ability to seek and obtain the regulatory approval required to commence product sales and generate revenue, which could cause our value to decline and limit our ability to obtain additional financing if needed.
| 49 |
Fast Track designation by the FDA for PF614 for chronic pain may not lead to a faster development or regulatory review or approval process and does not assure FDA approval.
We have obtained Fast Track designation for PF614 for management of moderate to severe chronic pain when a continuous, around-the-clock analgesic is needed for an extended period. We believe that Fast Track designation will enable us to facilitate the development and expedite the review of PF614. Fast Track designation does not ensure that PF614 will receive marketing approval or that approval will be granted within any timeframe. As a result, we may not experience a faster development process, review or approval compared to conventional FDA procedures. In addition, the FDA may withdraw Fast Track designation if it believes that the designation is no longer supported by data from our clinical development program. Fast Track designation does not guarantee that an NDA will obtain priority review designation. If any of these events occur, it could require us to conduct more extensive clinical trials and go through more extensive FDA reviews, which could substantially increase expenses and delay the time for commercializing our products.
If the FDA does not conclude that certain of our product candidates satisfy the requirements for the Section 505(b)(2) regulatory approval pathway, or if the requirements for such product candidates under Section 505(b)(2) are not as we expect, the approval pathway for those product candidates will likely take significantly longer, cost significantly more and entail significantly greater complications and risks than anticipated, and in either case may not be successful.
We may seek FDA approval through the Section 505(b)(2) regulatory pathway for our product candidate PF614. Section 505(b)(2) of the Federal Food, Drug and Cosmetic Act, or FDC Act, permits the submission of an NDA where at least some of the information required for approval comes from studies that were not conducted by or for the applicant and for which the applicant has not obtained a right of reference. Section 505(b)(2), if applicable to us under the FDC Act, would allow an NDA we submit to FDA to rely in part on data in the public domain or on the FDA’s prior conclusions regarding the safety and effectiveness of an approved product, or listed drug, which could expedite the development program for our product candidates by potentially decreasing the amount of data that we would need to generate in order to obtain FDA approval. If the FDA does not agree that the 505(b)(2) regulatory pathway is appropriate or scientifically justified for PF614, we may need to conduct additional preclinical and clinical trials, provide additional data and information, and meet additional standards for regulatory approval. For example, the FDA may not agree that we have provided a scientific bridge, through comparative bioavailability data, to demonstrate that reliance on the prior findings of safety or efficacy for a listed drug is justified. If this were to occur, the time and financial resources required to obtain FDA approval for this product candidate, and complications and risks associated with this product candidate, would likely substantially increase. We could need to obtain additional funding, which could result in significant dilution to the ownership interests of our then existing stockholders to the extent we issue equity securities or convertible debt. We cannot assure you that we would be able to obtain such additional financing on terms acceptable to us, if at all. Moreover, the inability to pursue the Section 505(b)(2) regulatory pathway may result in new competitive products reaching the market more quickly than our product candidates, which would likely materially adversely impact of our competitive position and prospects. Even if we are allowed to pursue the Section 505(b)(2) regulatory pathway, we cannot assure our stockholders that our product candidates will receive the requisite approvals for commercialization.
In addition, notwithstanding the approval of a number of products by the FDA under Section 505(b)(2) over the last few years, certain brand-name pharmaceutical companies and others have objected to the FDA’s interpretation of Section 505(b)(2). If the FDA’s interpretation of Section 505(b)(2) is successfully challenged, the FDA may change its 505(b)(2) policies and practices, which could delay or even prevent the FDA from approving any NDA that we submit under Section 505(b)(2). The pharmaceutical industry is highly competitive, and Section 505(b)(2) NDAs are subject to special requirements designed to protect the patent rights of sponsors of previously approved drugs that are referenced in a Section 505(b)(2) NDA. These requirements may give rise to patent litigation and mandatory delays in approval of our NDAs for up to 30 months or longer depending on the outcome of any litigation. It is not uncommon for a manufacturer of an approved product to file a citizen petition with the FDA seeking to delay approval of, or impose additional approval requirements for, pending competing products. If successful, such petitions can significantly delay, or even prevent, the approval of the new product. Even if the FDA ultimately denies such a petition, the FDA may substantially delay approval while it considers and responds to the petition. In addition, even if we are able to utilize the Section 505(b)(2) regulatory pathway, there is no guarantee this would ultimately lead to accelerated product development or earlier approval.
| 50 |
Moreover, even if our product candidates are approved under Section 505(b)(2), the approval may be subject to limitations on the indicated uses for which the products may be marketed or to other conditions of approval, or may contain requirements for costly post-marketing testing and surveillance to monitor the safety or efficacy of the products.
If we submit a 505(b)(2) application that references a third-party product, we may be subject to a patent infringement suit and the approval of our product may be delayed.
If we submit a 505(b)(2) application that relies in whole or in FDA’s findings for a listed drug, we will be required to certify to the FDA that either: (1) there is no patent information listed in the FDA’s publication Approved Drug Products with Therapeutic Equivalence Evaluations, which we refer to as the Orange Book, with respect to the listed drug; (2) the patents listed in the Orange Book have expired; (3) the listed patents have not expired, but will expire on a particular date and approval is sought after patent expiration; or (4) the listed patents are invalid or will not be infringed by the manufacture, use or sale of our product. A certification that our new drug will not infringe the Orange Book-listed patents for the applicable listed drug, or that such patents are invalid, is called a paragraph IV certification. If we submit a paragraph IV certification to the FDA, a notice of the paragraph IV certification must also be sent to the NDA holder once our 505(b)(2) application is filed by the FDA. The third party may then initiate a lawsuit to defend the patents identified in the notice. The filing of a patent infringement lawsuit within 45 days of receipt of the notice automatically prevents the FDA from approving our 505(b)(2) application until the earliest of 30 months or the date on which the patent expires, the lawsuit is settled, or the court reaches a decision in the infringement lawsuit in our favor. If the third party does not file a patent infringement lawsuit within the required 45-day period, our 505(b)(2) application will not be subject to the 30-month stay of FDA approval.
Changes in methods of product candidate manufacturing or formulation may result in additional costs or delay.
As product candidates proceed through preclinical studies to late-stage clinical trials towards potential approval and commercialization, it is common that various aspects of the development program, such as manufacturing methods and formulation, are altered along the way in an effort to optimize processes and results. Such changes carry the risk that they will not achieve these intended objectives. Any of these changes could cause our product candidates to perform differently and affect the results of planned clinical trials or other future clinical trials conducted with the materials manufactured using altered processes. Such changes may also require additional testing, FDA notification or FDA approval. This could delay or prevent completion of clinical trials, require conducting bridging clinical trials or repeating one or more clinical trials, increase clinical trial costs, delay or prevent approval of our product candidates and jeopardize our ability to commence sales and generate revenue.
Our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following regulatory approval, if obtained.
Undesirable side effects caused by any of our product candidates could cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in restrictive warnings or contraindication or the delay or denial of regulatory approval by the FDA or comparable foreign regulatory authorities. In our planned and future clinical trials of our product candidates, we may observe a less favorable safety and tolerability profile than was observed in earlier-stage testing of these candidates.
Undesirable side effects have been observed in our product candidates to date. For example, in clinical trials of PF614, opioid side effects were observed. Many compounds that initially showed promise in clinical or earlier-stage testing are later found to cause undesirable or unexpected side effects that prevented further development of the compound. Results of future clinical trials of our product candidates could reveal a high and unacceptable severity and prevalence of side effects or unexpected characteristics, despite a favorable tolerability profile observed in earlier-stage testing. If unacceptable side effects arise in the development of our product candidates, we, the FDA or comparable foreign regulatory authorities, the IRBs, or independent ethics committees at the institutions in which its trials are conducted, could suspend, limit or terminate our clinical trials, or the independent safety monitoring committee could recommend that we suspend, limit or terminate our trials, or the FDA or comparable foreign regulatory authorities could order us to cease clinical trials or deny approval of our product candidates for any or all targeted indications. Treatment-emergent side effects that are deemed to be drug-related could delay recruitment of clinical trial subjects or may cause subjects that enroll in our clinical trials to discontinue participation in our clinical trials. In addition, these side effects may not be appropriately recognized or managed by the treating medical staff. We may need to train medical personnel using our product candidates to understand the side effect profiles for our clinical trials and upon any commercialization of any of our product candidates. Inadequate training in recognizing or managing the potential side effects of our product candidates could result in harm to patients that are administered our product candidates. Any of these occurrences may adversely affect our business, financial condition and prospects significantly.
| 51 |
Moreover, clinical trials of our product candidates are conducted in carefully defined sets of patients who have agreed to enter into clinical trials. Consequently, it is possible that our clinical trials may indicate an apparent positive effect of a product candidate that is greater than the actual positive effect, if any, or alternatively fail to identify undesirable side effects.
FDA’s evolving standards for the approval of opioid products may delay or prevent approval of our product candidates.
FDA has identified addressing misuse and abuse of opioid drugs as one of its highest priorities. As part of its plan, the agency has established new standards for the development of prescription opioids with abuse-deterrent formulations and has published two sets of guidance. Since the publication of the second guidance in November 2017, FDA has not approved any new abuse–deterrent opioid drugs. If we are unable to meet FDA’s new and evolving standards for approving opioid products, we will not be able to market our products.
Product liability lawsuits against us or any of our future collaborators could divert our resources and attention, cause us to incur substantial liabilities and limit commercialization of our product candidates.
We are exposed to potential product liability and professional indemnity risks that are inherent in the research, development, manufacturing, marketing and use of pharmaceutical products. Currently, we have no products that have been approved for commercial sale; however, the use of our product candidates by us and any collaborators in clinical trials, and the sale of these product candidates, if approved, in the future, may expose us to liability claims. We face an inherent risk of product liability lawsuits related to the use of our product candidates in patients and will face an even greater risk if product candidates are approved by regulatory authorities and introduced commercially. Product liability claims may be brought against us by participants enrolled in our clinical trials, patients, health care providers, pharmaceutical companies, our collaborators or others using, administering or selling any of our future approved products. If we cannot successfully defend ourselves against any such claims, we may incur substantial liabilities or be required to limit commercialization of our product candidates. Regardless of the merits or eventual outcome, liability claims may result in:
| ● | decreased demand for any of our future approved products; | |
| ● | injury to our reputation; | |
| ● | withdrawal of clinical trial participants; | |
| ● | termination of clinical trial sites or entire trial programs; | |
| ● | significant litigation costs; | |
| ● | substantial monetary awards to, or costly settlements with, patients or other claimants; | |
| ● | product recalls or a change in the indications for which they may be used; | |
| ● | loss of revenue; | |
| ● | diversion of management and scientific resources from our business operations; and | |
| ● | the inability to commercialize our product candidates. |
Although the clinical trial process is designed to identify and assess potential side effects, clinical development does not always fully characterize the safety and efficacy profile of a new medicine, and it is always possible that a drug, even after regulatory approval, may exhibit unforeseen side effects. If our product candidates were to cause adverse side effects during clinical trials or after approval, we may be exposed to substantial liabilities. Physicians and patients may not comply with any warnings that identify known potential adverse effects and patients who should not use our product candidates. If any of our product candidates are approved for commercial sale, we will be highly dependent upon consumer perceptions of us and the safety and quality of our products. We could be adversely affected if we are subject to negative publicity associated with illness or other adverse effects resulting from patients’ use or misuse of our products or any similar products distributed by other companies.
| 52 |
Although we maintain product liability insurance coverage consistent with industry norms, including clinical trial liability, this insurance may not fully cover potential liabilities that we may incur. The cost of any product liability litigation or other proceeding, even if resolved in our favor, could be substantial. We will need to increase our insurance coverage if we commercialize any product that receives regulatory approval. In addition, insurance coverage is becoming increasingly expensive. If we are unable to maintain sufficient insurance coverage at an acceptable cost or to otherwise protect against potential product liability claims, it could prevent or inhibit the development and commercial production and sale of our product candidates, which could harm our business, financial condition, results of operations and prospects.
Oxycodone is a Schedule II controlled substance under the federal CSA, and any failure to comply with the CSA or its state equivalents would have a negative impact on our business.
Oxycodone, the ingredient in PF614, is classified as a Schedule II controlled substance under the Controlled Substances Act, or CSA and regulations promulgated by the DEA. The law and regulations classify substances as Schedule I, II, III, IV or V controlled substances, with Schedule I controlled substances considered to present the highest risk of substance abuse and Schedule V controlled substances the lowest risk. Scheduled controlled substances are subject to DEA regulations relating to supply, procurement, manufacturing, storage, shipment, sale, use, distribution and physician prescription procedures. For example, Schedule II controlled substances are subject to various restrictions, including, but not limited to, mandatory written prescriptions and the prohibition of refills. In addition to federal scheduling, oxycodone is subject to state-controlled substance laws and regulations, and in some cases, with additional requirements than those imposed by federal law and regulations. Though state controlled substances laws often mirror federal law, because the states are separate jurisdictions, they may schedule products separately.
Entities must register annually with the DEA to manufacture, distribute, dispense, import, export and conduct research using controlled substances. In addition, the DEA requires entities handling controlled substances to maintain complete and accurate records and file reports, including reports related to thefts or losses of any controlled substances, and to obtain authorization to destroy any controlled substances. Registered entities also must follow specific labeling and packaging requirements. Facilities must maintain appropriate security measures to control against diversion of controlled substances. Security requirements vary by controlled substance schedule with the most stringent requirements applying to Schedule I and Schedule II controlled substances. Required security measures include background checks on employees and physical control of inventory through measures such as vaults and inventory reconciliations.
Our contract manufacturing organizations, or CMOs, who manufacture and distribute PF614 are required to be registered with DEA and relevant state authorities and comply with all security, recordkeeping and reporting requirements. Manufacturers and distributors are subject to routine inspections and audits by the DEA related to compliance with security, recordkeeping and reporting requirements. Failure to maintain the required registrations or to comply and follow these requirements can lead to significant civil and/or criminal penalties and possibly even lead to a revocation of a DEA registration to manufacture or distribute such products.
Manufacturing of oxycodone is subject to annual quotas that limit the amount of API and dosage forms that can be produced in any given year; the failure of our CMOs to obtain the necessary manufacturing and/or procurement quota would have a negative impact on our business.
The CSA and DEA regulations establish an annual aggregate production quota for Schedule I and II controlled substances, including oxycodone and other narcotic drugs. In addition, each manufacturer of active pharmaceutical ingredient, or API or dosage forms must obtain an individual manufacturing or production quota that limits the amount of product that a company can produce and/or distribute each year. The DEA allocates manufacturing quota issued to companies so as to not exceed the aggregate quota established for a given year. Moreover, companies must demonstrate the need for procurement quota based on expected demand and sales of the controlled substance the DEA requires the submission of substantial evidence of expected legitimate medical and scientific need for the drug product before assigning its aggregate production quotas, or manufacturing and procurement quotas to manufacturers. The DEA has decreased the aggregate quota for certain narcotic drugs, including oxycodone over the last five years. Also, in October 2018, Congress passed the SUPPORT Act which requires the DEA to consider potential diversion in establishing quotas for narcotic drugs which could lead to continued decreases in quota available to API manufacturers and dosage form manufacturers of these substances.
| 53 |
In future years, we may need greater amounts of controlled substances that are subject to the DEA’s quota system to sustain our development program. We may also need significantly greater amounts to implement our commercialization plans if the FDA approves our proposed formulations. If any of our manufacturers of API or dosage forms are unable to obtain the necessary annual quota to meet the research and development or commercial demand for PF614, our business would be negatively impacted. Any delay or refusal by the DEA in establishing a quota, a reduction in quota, or a failure to increase quota over time could delay or stop the clinical development or commercial sale of some of our products or product candidates. This could have a material adverse effect on our business, results of operations, financial condition and prospects.
Risks Related to our Intellectual Property
If we are unable to obtain and maintain patent protection for our products candidates, or if the scope of the patent protection obtained is not sufficiently broad, our competitors could develop and commercialize product candidates that are similar or identical to our product candidates, and our ability to successfully commercialize our product candidates may be adversely affected.
Our commercial success will depend, in part, on our ability to obtain and maintain patent protection in the United States and other countries with significant commercial markets with respect to our product candidates. We seek to protect our proprietary position by filing patent applications in the United States and abroad related to our product candidates that are important to our business, as appropriate. We cannot be certain that patents will be issued or granted with respect to applications that are currently pending or that we may apply for in the future with respect to one or more of our product candidates, or that issued or granted patents will not later be found to be invalid and/or unenforceable.
The patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. Although we may enter into non-disclosure and confidentiality agreements with parties who have access to patentable aspects of our research and development output, such as our employees, distribution partners, consultants, advisors and other third parties, any of these parties may breach the agreements and disclose such output before a patent application is filed, thereby jeopardizing our ability to seek patent protection.
The patent position of pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial value of our patent rights are highly uncertain. Our pending and future patent applications may not result in patents being issued, and even if issued, the patents may not meaningfully protect our product candidates, effectively prevent competitors and third parties from commercializing competitive products or otherwise provide us with any competitive advantage. Even if the patent applications that we own or licenses issue as patents, they may not issue in a form that will provide us with any meaningful protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage. For product candidates for which we do not hold or do not obtain composition of matter patents, competitors who obtain the requisite regulatory approval can offer products with the same composition as our product candidate so long as the competitors do not infringe any method patents that we may hold. Method patents protect the product when used or sold for the specified method. However, this type of patent protection can be more difficult to enforce and does not limit a competitor from making and marketing a product that is identical to our product candidate that is either labeled or marketed for an indication that is outside of the patented method, or for which there is a substantial use in commerce outside the patented method. Our competitors or other third parties may be able to circumvent our patents by developing similar or alternative products in a non-infringing manner.
Changes in either the patent laws, implementing regulations or interpretation of the patent laws in the United States and other countries may also diminish the value of our patents or narrow the scope of our patent protection. The laws of foreign countries may not protect our rights to the same extent as the laws of the United States, and many companies have encountered significant difficulties in protecting and defending such rights in foreign jurisdictions.
| 54 |
We cannot be certain that our patents and patent rights will be effective in protecting our product candidates and technologies. Failure to protect such assets may have a material adverse effect on our business, operations, financial condition and prospects.
We may face litigation from third parties claiming that our products or business infringe, misappropriate, or otherwise violate their intellectual property rights, or seeking to challenge the validity of our patents.
Our future success is also dependent in part on the strength of our intellectual property, trade secrets and know-how, which have been developed from years of research and development, and on our ability, and the ability of our future collaborators, to develop, manufacture, market and sell our product candidates, if approved, and use our proprietary technologies without alleged or actual infringement, misappropriation or other violation of the patents and other intellectual property rights of third parties.
We may be exposed to, or be threatened with, adversarial proceedings or additional future litigation by third parties regarding intellectual property rights with respect to our current and any future product candidates and technology, including interference or derivation proceedings, post grant review and inter partes review before the United States Patent and Trademark Office, or USPTO, or similar adversarial proceedings or litigation in other jurisdictions seeking to challenge the validity of our intellectual property rights, claiming that we have misappropriated the trade secrets of others, or claiming that our technologies, products or activities infringe the intellectual property rights of others.
There have been many lawsuits and other proceedings involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interferences, oppositions, post grant review, inter partes review and reexamination proceedings before the USPTO, and corresponding foreign patent offices. Numerous United States and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which we are developing product candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our product candidates may be subject to claims of infringement of the intellectual property rights of third parties.
We are aware of patents owned by third parties, including potential competitors, that are directed to compositions comprising a chemically modified opioid, such as oxycodone, which decreases the potential of the opioid to be abused or cause overdose and related methods of use. Third parties, including potential competitors, may assert infringement claims against us based on existing patents or patents that may be granted in the future including, perhaps, the aforementioned patents, regardless of their merit. There is a risk that third parties may choose to engage in litigation with us to enforce or to otherwise assert their patent rights against us.
Even if we believe such claims are without merit, a court of competent jurisdiction could hold that these third-party patents are valid, enforceable and infringed, and the holders of any such patents may be able to block our ability to commercialize such product candidate unless we obtain a license under the applicable patents, or until such patents expire or are finally determined to be invalid or unenforceable. Similarly, if any third-party patents were held by a court of competent jurisdiction to cover aspects of our compositions, formulations, or methods of treatment, prevention or use, the holders of any such patents may be able to block our ability to develop and commercialize the applicable product candidate unless we obtain a license or until such patent expires or is finally determined to be invalid or unenforceable. In either case, such a license may not be available on commercially reasonable terms, or at all. Even if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. Some claimants may have substantially greater resources than we do and may be able to sustain the costs of complex intellectual property litigation to a greater degree and for longer periods of time than we could. In addition, patent holding companies that focus solely on extracting royalties and settlements by enforcing patent rights may target us.
Furthermore, even in the absence of litigation, we may need to obtain licenses from third parties to advance our research or to enable the commercialization of our product candidates. We may fail to obtain any of these licenses at a reasonable cost or on reasonable terms, if at all. In such an event, we would be unable to further practice our technologies or develop and commercialize any of our product candidates at issue, which could harm our business and financial condition significantly.
| 55 |
Parties making claims against us may obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize one or more of our product candidates, if approved. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of management and employee time and resources from our business. Third parties making such claims may have the ability to dedicate substantially greater resources to these legal actions than us or our licensors or collaborators can. In the event of a successful claim of infringement, misappropriation or other violation against us, we may have to pay substantial damages, including treble damages and attorneys’ fees for willful infringement, pay royalties, redesign our infringing products or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure.
Patent litigation and other proceedings may also absorb significant management time. The cost to us of any patent litigation or other proceeding, even if resolved in our favor, could be substantial. During the course of any patent or other intellectual property litigation or other proceeding, there could be public announcements of the results of hearings, rulings on motions, and other interim proceedings or developments and if securities analysts or investors regard these announcements as negative, the perceived value of our product candidates or intellectual property could be diminished. Accordingly, the market price of our common stock may decline. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our business, ability to compete in the marketplace, financial condition, results of operations and growth prospects.
We may become involved in lawsuits to protect or enforce our patents or other intellectual property, which could be expensive, time consuming and unsuccessful.
Competitors may infringe, misappropriate or otherwise violate our patents, trademarks, copyrights or other intellectual property, or those of our licensors. To counter infringement, misappropriation, unauthorized use or other violations, we may be required to file legal claims, which can be expensive and time consuming and divert the time and attention of our management and scientific personnel.
There can be no assurances that we will be successful with respect to any litigation matters which may arise in the ordinary course of our business. Such a failure may have a material impact on our business, results of operations and financial condition in the future.
We may not be able to prevent, alone or with any future licensors, infringement, misappropriation or other violations of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us alleging that we infringe their patents. In addition, in a patent infringement proceeding, there is a risk that a court will decide that a patent of ours is invalid or unenforceable, in whole or in part, and that we do not have the right to stop the other party from using the invention at issue. There is also a risk that, even if the validity of such patents is upheld, the court will construe the patent’s claims narrowly or decide that we do not have the right to stop the other party from using the invention at issue on the grounds that our patents do not cover the invention. An adverse outcome in a litigation or proceeding involving our patents could limit our ability to assert our patents against those parties or other competitors and may curtail or preclude our ability to exclude third parties from making and selling similar or competitive products. Any of these occurrences could adversely affect our competitive business position, business prospects and financial condition. Similarly, if we assert trademark infringement claims, a court may determine that the marks we have asserted are invalid or unenforceable, or that the party against whom we have asserted trademark infringement has superior rights to the marks in question. In this case, we could ultimately be forced to cease use of such trademarks.
In any infringement, misappropriation or other intellectual property litigation, any award of monetary damages we receive may not be commercially valuable. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during litigation. Moreover, there can be no assurance that we will have sufficient financial or other resources to file and pursue such infringement claims, which typically last for years before they are concluded. Even if we ultimately prevail in such claims, the monetary cost of such litigation and the diversion of the attention of our management and scientific personnel could outweigh any benefit we receive as a result of the proceedings.
| 56 |
The expiration or loss of patent protection may adversely affect our future revenues and operating earnings.
We rely on patent, trademark, trade secret and other intellectual property protection in the discovery, development, manufacturing and sale of our product candidates. In particular, patent protection is important in the development and eventual commercialization of our product candidates. Patents covering our product candidates normally provide market exclusivity, which is important in order to improve the probability that our product candidates are able to become profitable.
Certain of our patents relating to PF614 will expire in 2030. In addition, certain of our patents relating to the use of nafamostat for treating respiratory diseases will expire in 2028. While we are seeking additional patent coverage which may protect the technology underlying these patents, there can be no assurances that such additional patent protection will be granted, or if granted, that these patents will not be infringed upon or otherwise held enforceable. Even if we are successful in obtaining a patent, patents have a limited lifespan. In the United States, the natural expiration of a utility patent is generally 20 years after it is filed. Various extensions may be available; however, the life of a patent, and the protection it affords, is limited. Without patent protection of our product candidates, we may be open to competition from generic versions of such methods and compositions.
If we do not obtain protection under the Hatch-Waxman Amendments by extending the patent term, our business may be harmed.
Our commercial success will largely depend on our ability to obtain and maintain patent and other intellectual property in the United States and other countries with respect to our product candidates. Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting our product candidates might expire before or shortly after such candidates begin to be commercialized. We expect to seek extensions of patent terms in the United States and, if available, in other countries where we are prosecuting patents.
Depending upon the timing, duration and specifics of FDA marketing approval of our product candidates, one or more of our United States patents may be eligible for limited patent term extension, or PTE, under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent restoration term of up to five years beyond the normal expiration of the patent as compensation for patent term lost during development and the FDA regulatory review process, which is limited to the approved indication (and potentially additional indications approved during the period of extension) covered by the patent. This extension is limited to only one patent that covers the approved product, the approved use of the product, or a method of manufacturing the product. If we are unable to extend the expiration date of our existing patents or obtain new patents with longer expiry dates, our competitors may be able to take advantage of our investment in development and clinical trials by referencing our clinical and preclinical data to obtain approval of competing products following our patent expiration and launch their product earlier than might otherwise be the case.
We may not be able to protect our intellectual property rights throughout the world, which could negatively impact our business.
Filing, prosecuting and defending patents covering our product candidates in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. Further, licensing partners may not prosecute patents in certain jurisdictions in which we may obtain commercial rights, thereby precluding the possibility of later obtaining patent protection in these countries. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop our own products and may also export infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States. These products may compete with our product candidates, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
| 57 |
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets and other intellectual property protection, particularly those relating to biotechnology products, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license. Furthermore, while we intend to protect our intellectual property rights in our expected significant markets, we cannot ensure that we will be able to initiate or maintain similar efforts in all jurisdictions in which we may wish to market our product candidates. Accordingly, our efforts to protect our intellectual property rights in such countries may be inadequate, which may have an adverse effect on our ability to successfully commercialize our product candidates in all of our expected significant foreign markets.
Additionally, the requirements for patentability may differ in certain countries, particularly developing countries. For example, unlike other countries, China has a heightened requirement for patentability, and specifically requires a detailed description of medical uses of a claimed drug. In India, unlike the United States, there is no link between regulatory approval of a drug and our patent status. Furthermore, generic or biosimilar drug manufacturers or other competitors may challenge the scope, validity or enforceability of us or our licensors’ patents, requiring us or our licensees or any future licensors to engage in complex, lengthy and costly litigation or other proceedings. In addition, certain countries in Europe and developing countries, including China and India, have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In those countries, We and our licensees or any future licensors may have limited remedies if patents are infringed or if we or our licensees or any future licensors are compelled to grant a license to a third party, which could materially diminish the value of those patents. This could limit our potential revenue opportunities. Accordingly, we and our licensees’ or any future licensors’ efforts to enforce intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we own or license.
Changes in European law have caused uncertainty about our European patent portfolio and may result in additional costs to us.
In 2012, the European Patent Package, or EU Patent Package, regulations were passed with the goal of providing for a single pan-European Unitary Patent, and a new European Unified Patent Court, or UPC, for litigation of European patents. The EU Patent Package was ratified in February 2023 and currently covers 17 member states. On June 1, 2023, all European patents, including those issued prior to ratification, will by default automatically fall under the jurisdiction of the UPC and allow for the possibility of obtaining pan-European injunctions, and further will be at risk of a central revocation proceeding at the UPC in participating UPC states. Under the EU Patent Package, patent holders are permitted to “opt out” of the UPC on a patent-by-patent basis during an initial seven year period after the EU Patent Package is ratified, with the proviso that an “opt-out” is no longer available for EP patents for which a revocation has been initiated before the UPC. Owners of European patent applications who receive notice of grant after the EU Patent Package is ratified could, for the UPC contracting states, either obtain a Unitary Patent or validate the patent nationally and file an opt-out demand. The EU Patent Package may increase the uncertainties and costs surrounding the enforcement or defense of our issued European patents and pending applications. The full impact on future European patent filing strategy and the enforcement or defense of our issued European patents in member states and/or the UPC is not known.
| 58 |
We may be subject to claims that we or our employees, consultants, contractors or advisors have infringed, misappropriated or otherwise violated the intellectual property of a third party, or claiming ownership of what we regard as our own intellectual property.
Many of the contributors to our intellectual property, including patents and applications, were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees do not use the intellectual property and other proprietary information, know-how or trade secrets of others in their work for us, we may be subject to claims that we or these employees have used or disclosed such intellectual property or other proprietary information. Litigation may be necessary to defend against these claims.
In addition, while we typically require our employees, consultants and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own. For example, we have not obtained assignments for certain patent applications relating to abuse-resistant amphetamines. To the extent that we fail to obtain such assignments, such assignments do not contain a self-executing assignment of intellectual property rights or such assignments are breached, we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property.
Our reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them or that our trade secrets will be misappropriated or disclosed and if we are unable to protect the confidentiality of our trade secrets, the value of our technology could be materially adversely affected and our business would be harmed.
In addition to seeking patents for some of our technology and products, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, in seeking to develop and maintain a competitive position. Because we expect to rely on third parties to manufacture our product candidates and we expect to collaborate with third parties on the development of our product candidates, we must, at times, share trade secrets with them. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, consultants, independent contractors, advisors, corporate collaborators, outside scientific collaborators, contract manufacturers, suppliers and other third parties. We also enter into confidentiality and invention or patent assignment agreements with employees and certain consultants. We also seek to preserve the integrity and confidentiality of our data, trade secrets and know-how by maintaining physical security of our premises and physical and electronic security of our information technology systems. Monitoring unauthorized uses and disclosures is difficult, and we do not know whether the steps we have taken to protect our proprietary technologies will be effective.
Since our inception, we have sought to contract with manufacturers to supply commercial quantities of pharmaceutical formulations and products. As a result, we have disclosed, under confidentiality agreements, various aspects of our technology with potential manufacturers and suppliers. We believe that these disclosures, while necessary for our business, may have resulted and may result in the attempt by potential manufacturers and suppliers to improperly assert ownership claims to our technology in an attempt to gain an advantage in negotiating manufacturing and supplier rights.
We cannot guarantee that our trade secrets and other proprietary and confidential information will not be disclosed or that competitors will not otherwise gain access to our trade secrets. Any party with whom we have executed such an agreement may breach that agreement and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts both within and outside the United States may be less willing or unwilling to protect trade secrets. Further, if any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent such third party, or those to whom they communicate such technology or information, from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor, our business and competitive position could be harmed.
| 59 |
Trade secrets and know-how can be difficult to protect as trade secrets and know-how will over time be disseminated within the industry through independent development, the publication of journal articles, and the movement of personnel skilled in the art from company to company or academic to industry scientific positions. If we fail to prevent material disclosure of the know-how, trade secrets and other intellectual property related to our technologies to third parties, we will not be able to establish or maintain a competitive advantage in our market, which could materially adversely affect our business, results of operations and financial condition. Even if we are able to adequately protect our trade secrets and proprietary information, our trade secrets could otherwise become known or could be independently discovered by our competitors. For example, we are aware that certain of our former employees founded Elysium Therapeutics, which appears to be developing orally administered abuse deterrent opioids. Additionally, competitors could purchase our products and attempt to replicate some or all of the competitive advantages we derive from our development efforts, design around our protected technology or develop their own competitive technologies that fall outside of our intellectual property rights. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, in the absence of patent protection, we would have no right to prevent them, or those to whom they communicate, from using that technology or information to compete with us.
We may not be able to prevent misappropriation of our intellectual property, trade secrets or confidential information, particularly in countries where the laws may not protect those rights as fully as in the United States. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation.
We may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent, which might adversely affect our ability to develop and market our product candidates.
To the extent undertaken, we cannot guarantee that any of our patent searches or analyses, including the identification of relevant patents, the scope of patent claims or the expiration of relevant patents, are complete or thorough, nor can we be certain that we have identified each and every third-party patent and pending application in the United States and abroad that is or may be relevant to or necessary for the commercialization of our product candidates in any jurisdiction. Patent applications in the United States and elsewhere are not published until approximately 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. In addition, certain United States patent applications can remain confidential until patents issue. Therefore, patent applications covering our products could have been filed by others without our knowledge. Additionally, pending patent applications that have been published can, subject to certain limitations, be later amended in a manner that could cover our product candidates or the use of our product candidates.
The scope of a patent claim is determined by an interpretation of the law, the written disclosure in a patent and the patent’s prosecution history. Our interpretation of the relevance or the scope of a patent or a pending application may be incorrect, which may negatively impact our ability to market our product candidates. We may incorrectly determine that our product candidates are not covered by a third-party patent or may incorrectly predict whether a third party’s pending application will issue with claims of relevant scope. Our determination of the expiration date of any patent in the United States or abroad that we consider relevant may be incorrect, and our failure to identify and correctly interpret relevant patents may negatively impact our ability to develop and market our product candidates.
If we fail to identify and correctly interpret relevant patents, we may be subject to infringement claims. We cannot guarantee that we will be able to successfully settle or otherwise resolve such infringement claims. If we fail in any such dispute, in addition to being forced to pay damages, we may be temporarily or permanently prohibited from commercializing any of our product candidates that are held to be infringing. We might, if possible, also be forced to redesign product candidates or services so that we no longer infringe the third-party intellectual property rights. Any of these events, even if we were ultimately to prevail, could require us to divert substantial financial and management resources that we would otherwise be able to devote to our business.
| 60 |
Intellectual property rights do not necessarily address all potential threats to our business.
Once granted, patents may remain open to opposition, interference, re-examination, post-grant review, inter partes review, nullification or derivation action in court or before patent offices or similar proceedings for a given period after allowance or grant, during which time third parties can raise objections against such grant. In the course of such proceedings, which may continue for a protracted period of time, the patent owner may be compelled to limit the scope of the allowed or granted claims thus attacked or may lose the allowed or granted claims altogether. In addition, the degree of future protection afforded by our intellectual property rights is uncertain because even granted intellectual property rights have limitations and may not adequately protect our business. The following examples are illustrative:
| ● | others may be able to make formulations that are similar to our product candidates or other formulations but that are not covered by the claims of our patent rights; | |
| ● | the patents of third parties may have an adverse effect on our business; | |
| ● | we or any future strategic partners might not have been the first to conceive or reduce to practice the inventions covered by the issued patent or pending patent application that we own; | |
| ● | we or any future strategic partners might not have been the first to file patent applications covering certain of our inventions; | |
| ● | others may independently develop similar or alternative technologies or duplicate any of our technologies without infringing our intellectual property rights; | |
| ● | it is possible that our pending patent applications will not lead to issued patents; | |
| ● | issued patents that we may own or that we exclusively license in the future may not provide us with any competitive advantage, or may be held invalid or unenforceable, as a result of legal challenges by our competitors; | |
| ● | our competitors might conduct research and development activities in countries where we do not have patent rights and then use the information learned from such activities to develop competitive products for sale in our major commercial markets; | |
| ● | third parties performing manufacturing or testing for us using our product candidates or technologies could use the intellectual property of others without obtaining a proper license; | |
| ● | we may not develop additional proprietary technologies that are patentable; and | |
| ● | the patents of others may have an adverse effect on our business. |
Should any of these events occur, they could have a material adverse effect on our business, financial condition, results of operations and prospects.
The validity, scope and enforceability of any patents listed in the Orange Book that cover our product candidates can be challenged by third parties.
If one of our product candidates is approved by the FDA, one or more third parties may challenge the current patents, or patents that may issue in the future, within our portfolio which could result in the invalidation of, or render unenforceable, some or all of the relevant patent claims or a finding of non-infringement. For example, if a third party submits an application under Section 505(b)(2) or an abbreviated new drug application, or ANDA, for a generic drug containing any of our product candidates, and relies in whole or in part on studies conducted by or for us, the third party will be required to certify to the FDA that either: (1) there is no patent information listed in the Orange Book with respect to our NDA for the applicable approved drug candidate; (2) the patents listed in the Orange Book have expired; (3) the listed patents have not expired, but will expire on a particular date and approval is sought after patent expiration; or (4) the listed patents are invalid or will not be infringed by the manufacture, use or sale of the third party’s generic drug. A certification that the new drug will not infringe the Orange Book-listed patents for the applicable approved drug candidate, or that such patents are invalid, is called a paragraph IV certification. If the third party submits a paragraph IV certification to the FDA, a notice of the paragraph IV certification must also be sent to us once the third party’s ANDA is accepted for filing by the FDA. We may then initiate a lawsuit to defend the patents identified in the notice. The filing of a patent infringement lawsuit within 45 days of receipt of the notice automatically prevents the FDA from approving the third party’s ANDA until the earliest of 30 months or the date on which the patent expires, the lawsuit is settled, or the court reaches a decision in the infringement lawsuit in favor of the third party. If we do not file a patent infringement lawsuit within the required 45-day period, the third party’s ANDA will not be subject to the 30-month stay of FDA approval.
| 61 |
Moreover, a third party may challenge the current patents, or patents that may be issued in the future, within our portfolio which could result in the invalidation of some or all the patents that might otherwise be eligible for listing in the Orange Book for one of our products. If a third party successfully challenges all of the patents that might otherwise be eligible for listing in the Orange Book for one of our products, we will not be entitled to the 30-month stay of FDA approval upon the filing of an ANDA for a generic drug containing any of our product candidates, and relies in whole or in part on studies conducted by or for us. Litigation or other proceedings to enforce or defend intellectual property rights are often very complex in nature, may be very expensive and time-consuming, may divert our management’s attention from our core business, and may result in unfavorable results that could limit our ability to prevent third parties from competing with our product candidates.
Cyber-attacks or other failures in our telecommunications or information technology systems, or those of our collaborators, CROs, third-party logistics providers, distributors or other contractors or consultants, could result in information theft, data corruption and significant disruption of our business operations.
We, our collaborators, CROs, third-party logistics providers, distributors and other contractors and consultants utilize information technology, or IT, systems and networks to process, transmit and store electronic information in connection with our business activities. As use of digital technologies has increased, cyber incidents, including third parties gaining access to employee accounts using stolen or inferred credentials, computer malware, viruses, spamming, phishing attacks or other means, and deliberate attacks and attempts to gain unauthorized access to computer systems and networks, have increased in frequency and sophistication. These threats pose a risk to the security of us, our collaborators’, CROs’, third-party logistics providers’, distributors’ and other contractors’ and consultants’ systems and networks, and the confidentiality, availability and integrity of our data. There can be no assurance that we will be successful in preventing cyber-attacks or successfully mitigating their effects. Similarly, there can be no assurance that our collaborators, CROs, third-party logistics providers, distributors and other contractors and consultants will be successful in protecting our clinical and other data that is stored on their systems. Like other companies, we have on occasion experienced, and will continue to experience, threats to our data and systems, including malicious codes and viruses, phishing, business email compromise attacks or other cyber-attacks. Any cyber-attack, data breach or destruction or loss of data could result in a violation of applicable United States and international privacy, data protection and other laws and subject us to litigation and governmental investigations and proceedings by federal, state and local regulatory entities in the United States and by international regulatory entities, resulting in exposure to material civil and/or criminal liability. Further, our general liability insurance and corporate risk program may not cover all potential claims to which we are exposed and may not be adequate to indemnify us for all liability that may be imposed, which could have a material adverse effect on our business and prospects. For example, the loss of clinical trial data from completed or ongoing clinical trials for any of our product candidates could result in delays in our development and regulatory approval efforts and significantly increase our costs to recover or reproduce the data. In addition, we may suffer reputational harm or face litigation or adverse regulatory action as a result of cyber-attacks or other data security breaches and may incur significant additional expense to implement further data protection measures.
Risks Related to the Ownership of Common Stock and Financial Reporting
Raising additional capital has caused, and may in the future cause, dilution to our stockholders, adversely affect the market price of our common stock, restrict our operations or require us to relinquish rights to our technologies or product candidates.
We expect our expenses to increase in connection with our planned operations. Unless and until we can generate a substantial amount of revenue from our product candidates, we expect to finance our future cash needs through public or private equity offerings, debt financings, collaborations, licensing arrangements or other sources, or any combination of the foregoing. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe that we have sufficient funds for our current or future operating plans.
| 62 |
To the extent that we raise additional capital through the sale of common stock, convertible securities or other equity securities as we have done in the past, our stockholders’ ownership interest has been, and may in the future be, diluted. In addition, debt financing, if available, may result in fixed payment obligations and may involve agreements that include restrictive covenants that limit our ability to take specific actions, such as incurring additional debt, making capital expenditures, creating liens, redeeming stock or declaring dividends, that could adversely impact our ability to conduct our business. In addition, securing financing could require a substantial amount of time and attention from our management and may divert a disproportionate amount of their attention away from day-to-day activities, which may adversely affect our management’s ability to oversee the development of our product candidates.
In addition, we may sell securities in the public or private equity markets if and when conditions are favorable, or at prices per share below the current market price of our common stock, even if we do not have an immediate need for additional capital at that time. Sales of substantial amounts of shares of our common stock, or the perception that such sales could occur, could adversely affect the prevailing market price of our shares and our ability to raise capital. We may issue additional shares of common stock in future financing transactions or as incentive compensation for our executive management and other key personnel, consultants and advisors. Moreover, sales of substantial amounts of shares in the public market, or the perception that such sales could occur, may adversely affect the prevailing market price of our common stock and make it more difficult for us to raise additional capital.
Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making acquisitions, engaging in acquisition, merger or collaboration transactions, selling or licensing our assets, making capital expenditures, redeeming our stock, making certain investments, declaring dividends or encumbering our assets to secure future indebtedness. Such restrictions could adversely impact our ability to conduct our operations and execute our business plan. The Investor Notes contain such restrictions including a pledge of substantially all of our tangible and intangible assets, including our intellectual property.
If we raise additional funds through upfront payments or milestone payments pursuant to strategic collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies or intellectual property, future revenue streams, research programs or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
We are a smaller reporting company within the meaning of the Securities Act, and if we take advantage of certain exemptions from disclosure requirements available to “smaller reporting companies,” this could make our securities less attractive to investors and may make it more difficult to compare our performance with other public companies.
We are a “smaller reporting company” as defined in Item 10(f)(1) of Regulation S-K. Smaller reporting companies may take advantage of certain reduced disclosure obligations, including, among other things, providing only two years of audited financial statements. We will remain a smaller reporting company until the last day of the fiscal year in which (i) the market value of our common stock held by non-affiliates is greater than or equal to $250 million as of the end of that fiscal year’s second fiscal quarter, and (ii) our annual revenues are greater than or equal to $100 million during the last completed fiscal year and the market value of our common stock held by non-affiliates exceeds $700 million as of the end of that fiscal year’s second fiscal quarter. To the extent we take advantage of such reduced disclosure obligations, it may also make comparison of our financial statements with other public companies difficult or impossible.
| 63 |
Our internal controls over financial reporting currently may not meet all of the standards contemplated by Section 404 of Sarbanes-Oxley Act, and failure to achieve and maintain effective internal controls over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act could impair our ability to produce timely and accurate financial statements or comply with applicable regulations and have a material adverse effect on our business.
Our management has significant requirements for enhanced financial reporting and internal controls as a public company. The process of designing and implementing effective internal controls is a continuous effort that will require us to anticipate and react to changes in our business and the economic and regulatory environments and to expend significant resources to maintain a system of internal controls that is adequate to satisfy our reporting obligations as a public company. If we are unable to maintain appropriate internal financial reporting controls and procedures, it could cause us to fail to meet our reporting obligations on a timely basis or result in material misstatements in our consolidated financial statements, which could harm our operating results. In addition, we are required, pursuant to Section 404 of the Sarbanes-Oxley Act, to furnish a report by management on, among other things, the effectiveness of our internal controls over financial reporting. This assessment needs to include disclosure of any material weaknesses identified by our management in our internal control over financial reporting. The rules governing the standards that must be met for our management to assess our internal control over financial reporting are complex and require significant documentation, testing, and possible remediation.
Matters impacting our internal controls may cause us to be unable to report our financial information on a timely basis and thereby subject us to adverse regulatory consequences, including sanctions by the SEC, or violations of applicable stock exchange listing rules, which may result in a breach of the covenants under existing or future financing arrangements. There also could be a negative reaction in the financial markets due to a loss of investor confidence in us and the reliability of our financial statements. This could materially adversely affect us and lead to a decline in the market price of our common stock.
The amount of our future losses is uncertain and our quarterly and annual operating results may fluctuate significantly or fall below the expectations of investors or securities analysts, each of which may cause our stock price to fluctuate or decline.
Our quarterly and annual operating results may fluctuate significantly in the future due to a variety of factors, many of which are outside of our control and may be difficult to predict, including the following:
| ● | the timing and success or failure of clinical trials for our product candidates or competing product candidates, or any other change in the competitive landscape of our industry, | |
| ● | our ability to successfully recruit and retain subjects for clinical trials, and any delays caused by difficulties in such efforts; | |
| ● | the risk/benefit profile, cost and reimbursement policies with respect to our product candidates, if approved, and existing and potential future therapeutics that compete with our product candidates; | |
| ● | our ability to obtain marketing approval for our product candidates and the timing and scope of any such approvals we may receive; | |
| ● | the timing and cost of, and level of investment in, research and development activities relating to our product candidates, which may change from time to time; | |
| ● | the cost of manufacturing our product candidates, which may vary depending on the quantity of production and the terms of our agreements with manufacturers; | |
| ● | our ability to attract, hire and retain qualified personnel; | |
| ● | expenditures that we will or may incur to develop additional product candidates; | |
| ● | the level of demand for our product candidates should they receive approval, which may vary significantly; | |
| ● | the changing and volatile U.S. and global economic environments; and | |
| ● | future accounting pronouncements or changes in our accounting policies. |
| 64 |
The cumulative effects of these factors could result in large fluctuations and unpredictability in our quarterly and annual operating results. As a result, comparing our operating results on a period-to-period basis may not be meaningful. This variability and unpredictability could also result in our failing to meet the expectations of industry or financial analysts or investors for any period. If our operating results or revenue fall below the expectations of analysts or investors or below any forecasts we may provide to the market, or if the forecasts we provide to the market are below the expectations of analysts or investors, the price of our common stock could decline substantially. Such a stock price decline could occur even when we have met any previously publicly stated guidance we may provide, if any.
Warrants for shares of our common stock, if exercised, will increase the number of shares eligible for future resale in the public market and result in dilution to our stockholders.
We have issued warrants to purchase shares of our common stock in connection with various financing transactions. To the extent these warrants are exercised, additional shares of our common stock will be issued, which will result in dilution to the holders of shares of our common stock and increase the number of shares of common stock eligible for resale in the public market. Sales of substantial numbers of such shares of common stock in the public market or the fact that such warrants may be exercised could adversely affect the market price of our common stock.
Our issuance of additional capital stock in connection with financings, acquisitions, investments and our 2021 Omnibus Incentive Plan will dilute all other stockholders.
We expect to issue additional capital stock in the future that will result in dilution to all other stockholders. We have granted equity awards to employees, directors, and consultants under our 2021 Omnibus Incentive Plan and plan to do so in the future. We may also raise capital through equity financings in the future. As part of our business strategy, we may acquire or make investments in complementary companies, products, or technologies and issue equity securities to pay for any such acquisition or investment. Any such issuances of additional capital stock may cause stockholders to experience significant dilution of their ownership interests and the per share value of our common stock to decline.
If we are unable to regain compliance with the listing standards of Nasdaq, our common stock could be delisted and may become subject to “penny stock” rules, which could have a material adverse effect on the liquidity of our common stock, the ability of investors to sell their shares and our ability to raise funding.
On November 13, 2023, we received notice from the Listing Qualifications department of Nasdaq stating that, due to our non-compliance with the $2.5 million stockholders’ equity requirement set forth in Nasdaq Listing Rule 5550(b)(1) as of September 30, 2023, we were subject to delisting unless we timely request a hearing before the Nasdaq Hearings Panel (the “Panel”), which we did, resulting in a hearing before the Panel in early February 2024. The Panel granted our request for continued listing until May 13, 2024. We intend to actively explore options to regain compliance with Nasdaq listing requirements; however, there can be no assurance that we will be able to evidence compliance prior to the expiration of the extension granted to us by the Panel.
The de-listing of our common stock on Nasdaq could have a material adverse effect on us, including on our ability to raise capital through alternative financing sources on terms acceptable to us, or at all, and may result in the potential loss of confidence by investors, suppliers, customers and employees and fewer business development opportunities. Upon any delisting, our common stock could become subject to the regulations of the SEC relating to the market for penny stocks. Penny stocks are securities with a price of less than $5.00 per share unless (i) the securities are traded on a “recognized” national exchange or (ii) the issuer has net tangible assets less than $2,000,000 (if the issuer has been in continuous operation for at least three years) or $5,000,000 (if in continuous operation for less than three years), or with average annual revenues of less than $6,000,000 for the last three years.
The procedures applicable to penny stocks requires a broker-dealer to (i) obtain from the investor information concerning his financial situation, investment experience and investment objectives; (ii) reasonably determine, based on that information, that transactions in penny stocks are suitable for the investor and that the investor has sufficient knowledge and experience as to be reasonably capable of evaluating the risks of penny stock transactions; (iii) provide the investor with a written statement setting forth the basis on which the broker-dealer made the determination in (ii) above; and (iv) receive a signed and dated copy of such statement from the investor, confirming that it accurately reflects the investor’s financial situation, investment experience and investment objectives. The regulations applicable to penny stocks may severely affect the market liquidity for our common stock and could limit the ability of stockholders to sell their common stock in the secondary market.
| 65 |
Item 1B. Unresolved Staff Comments
Not applicable.
Item 1C. Cybersecurity
Risk Management and Strategy
We have developed and implemented cybersecurity risk management processes intended to protect the confidentiality, integrity and availability of our critical systems and information.
Our cybersecurity risk management program includes:
| ● | physical, technological and administrative controls intended to support our cybersecurity and data governance framework, including protections designed to protect the confidentiality, integrity and availability of our key information systems; | |
| ● | risk assessments designed to help identify material cybersecurity risks to our critical systems and information; | |
| ● | designated team members are responsible for managing our cybersecurity risk assessment processes, our security controls, and our response to cybersecurity incidents; and | |
| ● | the use of external service providers, where appropriate, to assess, test or otherwise assist with aspects of our security controls. |
We have not identified risks from known cybersecurity threats that have materially affected or are reasonably likely to materially affect us, including our operations, business strategy, results of operations, or financial condition. See Item 1A. “Risk Factors – Cyber-attacks or other failures in our telecommunications or information technology systems, or those of our collaborators, CROs, third-party logistics providers, distributors or other contractors or consultants, could result in information theft, data corruption and significant disruption of our business operations.”
Governance
Our Board considers cybersecurity risk as part of its risk oversight function. The Audit Committee oversees our cybersecurity and other information technology risks and management’s implementation of our cybersecurity risk management program. The Audit Committee receives periodic reports from management on our cybersecurity risks. In addition, management updates the Board and the Audit Committee, as necessary, regarding any material cybersecurity incidents, as well as any incidents with lesser impact potential.
Our management team, including our Chief Financial Officer, is responsible for assessing and managing our material risks from cybersecurity threats. Our Chief Financial Officer has primary responsibility for our overall cybersecurity risk management program. Our Chief Financial Officer has experience supervising and managing company information technology departments.
Our management team supervises efforts to prevent, detect, mitigate, and remediate cybersecurity risks and incidents through various means, which may include briefings from external security personnel; threat intelligence and other information obtained from governmental, public or private sources; and alerts and reports produced by security tools deployed in the information technology environment.
Item 2. Properties
Our principal executive office is located at 7946 Ivanhoe Ave., Suite 201 in La Jolla, California, where we lease a total of 850 square feet of office space that we use for our administrative activities. All development activities are undertaken at contract research organizations. The lease expires in October 2024. We believe that our current arrangements will be sufficient to meet our needs for the foreseeable future, and that, should it be needed, suitable space will be available to accommodate our administrative activities.
| 66 |
Item 3. Legal Proceedings
From time to time, we could become involved in disputes and various litigation matters that arise in the normal course of business. These may include disputes and lawsuits related to intellectual property, licensing, contract law and employee relations matters. Periodically, we review the status of significant matters, if any exist, and assess the potential financial exposure. If the potential loss from any claim or legal claim is considered probable and the amount can be estimated, we accrue a liability for the estimated loss. Legal proceedings are subject to uncertainties, and the outcomes are difficult to predict. Because of such uncertainties, accruals are based on the best information available at the time. As additional information becomes available, we reassess the potential liability related to pending claims and litigation. As of the date hereof, we are not a party to any material legal proceeding.
Item 4. Mine Safety Disclosures
Not applicable.
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Price and Ticker Symbol
Our common stock is currently listed on the Nasdaq Stock Market under the symbol “ENSC.” Our Public Warrants are currently listed on the OTC Pink Open Market under the symbol “ENSCW.”
The closing price of our common stock and Public Warrants on March 8, 2024, was $0.7852 and $0.041, respectively.
Holders
As of March 8, 2024, there were approximately 71 holders of record of our common stock.
Such numbers do not include beneficial owners holding our securities through nominee names.
Dividends
We have not paid any cash dividends on our common stock to date. We may retain future earnings, if any, for future operations, expansion and debt repayment and has no current plans to pay cash dividends for the foreseeable future. Any decision to declare and pay dividends in the future will be made at the discretion of the Board and will depend on, among other things, our results of operations, financial condition, cash requirements, contractual restrictions and other factors that the Board may deem relevant. In addition, our ability to pay dividends may be limited by covenants of any existing and future outstanding indebtedness we or our subsidiaries incur. We do not anticipate declaring any cash dividends to holders of our common stock in the foreseeable future.
| 67 |
Securities Authorized for Issuance under Equity Compensation Plans
The following table provides information as of December 31, 2023, with respect to securities that may be issued under our equity compensation plans:
| Plan Category | Number of Securities to be Issued upon Exercise of Outstanding Options, Warrants and Rights | Weighted Average Exercise Price of Outstanding Options, Warrants and Rights | Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (Excluding Securities Reflected in the First Column) | |||||||||
| Equity compensation plans approved by security holders | 581,314 | $ | 33.15 | 2,112 | ||||||||
| Equity compensation plans not approved by security holders | - | - | - | |||||||||
| Total | 581,314 | $ | 33.15 | 2,112 | ||||||||
Recent Sales of Unregistered Securities and Use of Proceeds
On October 23, 2023, we entered into a Securities Purchase Agreement for an aggregate financing of $1.7 million with investors. The Company issued to the investors (i) 2023 Notes in the aggregate principal amount of $1.8 million for an aggregate purchase price of $1.7 million and (ii) warrants to purchase 3.8 million shares of the Company’s common stock in the aggregate at an exercise price of $1.5675 per share. The first funding of $0.6 million occurred on October 25, 2023 and the second funding of $1.1 million occurred on November 28, 2023. Pursuant to the 2023 Notes, shares of Company common stock were issued to these investors in satisfaction of principal and interest payments. The proceeds are being used for working capital purposes subject to certain customary restrictions. See, “Liquidity and Capital Resources” for a detailed description of the 2023 Notes.
None of the foregoing transactions involved any underwriters, underwriting discounts or commissions, or any public offering. Unless otherwise set forth above, we believe each of these transactions was exempt from registration under the Securities Act in reliance on Section 4(a)(2) of the Securities Act (and Regulation D promulgated thereunder) as transactions by an issuer not involving any public offering or Rule 701 promulgated under Section 3(b) of the Securities Act as transactions by an issuer under benefit plans and contracts relating to compensation as provided under Rule 701. The recipients of the securities in each of these transactions represented their intentions to acquire the securities for investment only and not with a view to or for sale in connection with any distribution thereof, and appropriate legends were placed on the share certificates issued in these transactions. All recipients had adequate access, through their relationships with us, to information about us. The sales of these securities were made without any general solicitation or advertising.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
| 68 |
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
Overview
Ensysce is a clinical stage pharmaceutical company seeking to develop innovative solutions for severe pain relief while reducing the fear of and the potential for opioid misuse, abuse and overdose. Our lead product candidate, PF614, is an extended release TAAP prodrug of oxycodone. TAAP modification of prescription drugs removed the ability to crush, chew or manipulate and inject to achieve the effect of the medication more quickly than by swallowing. MPAR® adds a layer of overdose protection to each TAAP product.
Since our inception in 2003, we devoted substantially all of our efforts and financial resources to organizing and staffing our company, business planning, raising capital, discovering product candidates and securing related intellectual property rights and conducting research and development activities for our product candidates. We do not have any products approved for sale and we have not generated any revenue from product sales. We may never be able to develop or commercialize a marketable product.
Our lead product candidate, PF614, is in Phase 2 clinical development, PF614-MPAR is in Phase 1b clinical development and nafamostat is proceeding towards Phase 2 clinical development. Our other product candidates and our research initiatives are in preclinical or earlier stages of development. Our ability to generate revenue from product sales sufficient to achieve profitability will depend heavily on the successful development and eventual commercialization of one or more of our product candidates. We have not yet successfully completed any pivotal clinical trials, nor have we obtained any regulatory approvals, manufactured a commercial-scale drug, or conducted sales and marketing activities.
We have incurred significant operating losses since inception and we expect to continue to incur net losses for the foreseeable future. We expect that our expenses and capital requirements will increase substantially in connection with our ongoing development activities, particularly if and as we:
| ● | continue preclinical studies and continues existing and initiates new clinical trials for PF614, PF614-MPAR and nafamostat, our lead product candidates being tested for chronic pain and infectious disease; | |
| ● | advance the development of our product candidate pipeline of other product candidates, including through business development efforts to invest in or in-license other technologies or product candidates; | |
| ● | maintain, expand and protect our intellectual property portfolio; | |
| ● | hire additional clinical, quality control, medical, scientific and other technical personnel to support our clinical operations; | |
| ● | seek regulatory approval for any product candidates that successfully complete clinical trials; | |
| ● | undertake any pre-commercialization activities to establish sales, marketing and distribution capabilities for any product candidates for which we may receive regulatory approval; | |
| ● | expand our infrastructure and facilities to accommodate our growing employee base; and | |
| ● | add operational, financial and management information systems and personnel, including personnel to support our research and development programs, any future commercialization efforts and our transition to operating as a public company. |
We have incurred and expect to continue to incur additional costs associated with operating as a public company, including significant legal, accounting, insurance, investor relations and other expenses. We may never become profitable.
| 69 |
We require substantial additional funding to support our continuing operations and pursue our growth strategy. Until we can generate significant revenue from product sales, if ever, we expect to finance our operations through a combination of private and public equity offerings, debt financings or other capital sources, which may include collaborations with other companies or other strategic transactions. To the extent that we raise additional capital through the sale of private or public equity or convertible debt securities, existing ownership interests will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our equity holders.
Debt financing and equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, issuing additional equity, making acquisitions or capital expenditures or declaring dividends. If we raise additional funds through collaborations or other strategic transactions with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or drug candidates, or grant licenses on terms that may not be favorable to us. We may be unable to raise additional funds or enter into such other agreements or arrangements when needed on favorable terms, or at all. If we fail to raise capital or enter into such agreements as and when needed, we may have to significantly delay, scale back or discontinue the development and commercialization of one or more of our product candidates or delay our pursuit of potential in-licenses or acquisitions.
Because of the numerous risks and uncertainties associated with product development, we are unable to predict the timing or amount of increased expenses or when or if we will be able to achieve or maintain profitability. Even if we are able to generate product sales, we may not become profitable. If we fail to become profitable or are unable to sustain profitability on a continuing basis, we may be unable to continue our operations at planned levels and be forced to reduce or terminate our operations.
We have generated limited revenues and have incurred significant operating losses since our inception and expect to continue to incur operating losses for the foreseeable future. These factors raise substantial doubt about our ability to continue as a going concern. Our future viability is dependent on our ability to raise additional capital to finance our operations. Without raising additional capital through a future offering, we believe that current cash on hand is sufficient to fund operations into the third quarter of 2024. We based this estimate on assumptions that may prove to be wrong, and we could exhaust our available capital resources sooner than we expect. See “— Liquidity and Capital Resources.” Our future viability beyond the twelve months is dependent on our ability to raise additional capital to finance our operations.
We expect to incur substantial expenses in the foreseeable future for the development and potential commercialization of our product candidates and ongoing internal research and development programs. At this time, we cannot reasonably estimate the nature, timing or aggregate amount of costs for our development, potential commercialization, and internal research and development programs. However, in order to complete our current and future preclinical studies and clinical trials, and to complete the process of obtaining regulatory approval for our product candidates, as well as to build the sales, marketing and distribution infrastructure that we believe will be necessary to commercialize our product candidates, if approved, we may require substantial additional funding in the future.
2021 Notes
On September 24, 2021, we entered into the SPA for an aggregate financing of $15.0 million with institutional investors. A first closing under the SPA occurred on September 24, 2021 and a second closing under the SPA occurred on November 5, 2021. At the first closing, the Company issued to the investors (i) senior secured convertible promissory notes in the aggregate principal amount of $5.3 million for an aggregate purchase price of $5.0 million and (ii) warrants to purchase 1,507 shares of the Company’s common stock in the aggregate at a current exercise price of $3.64 per share. At the second closing, the Company issued to the institutional investors referenced above, (i) senior secured convertible promissory notes in the aggregate principal amount of $10.6 million for an aggregate purchase price of $10.0 million and (ii) warrants to purchase 3,011 shares of the Company’s common stock in the aggregate at a current exercise price of $3.64 per share. The 2021 Notes were satisfied on October 10, 2022.
| 70 |
2022 Notes
On June 30, 2022, we entered into an $8.0 million convertible financing agreement with institutional investors. The agreement provided for two closings, each for notes payable of $4.24 million (resulting in gross cash proceeds of $4.0 million). Funds were received for the first closing on July 1, 2022 and for the second closing on August 9, 2022. The remaining amount of principal and interest on the 2022 Notes was repaid in the first quarter of 2023. We were obligated under the 2022 Notes to pay additional cash as true-up payments for interest or redemption amounts that we paid in shares of common stock that were valued below $24.07 or the lower conversion price of $9.01 in effect between January 12, 2023 and May 12, 2023. The true-up payments compensate the holder for the difference between the value of a share and the conversion price in effect at the time of redemption, multiplied by the number of shares paid. The true-up payments totaling $0.6 million were paid on May 12, 2023.
In connection with each of the first and second closings of the 2022 Notes we also issued warrants to purchase 38,900 shares of the Company’s common stock. The warrants have a current exercise price of $3.64 and are exercisable for five years following issuance of the 2022 Notes.
2022 December Offering
On December 7, 2022, we entered into an underwriting agreement with Lake Street Capital Management, LLC (the “Underwriter”), pursuant to which we agreed to issue and sell (i) 190,000 shares of the Company’s common stock, par value $0.0001 per share, (ii) pre-funded warrants to purchase 51,666 shares of common stock and (iii) warrants to purchase 483,333 shares of common stock to the Underwriter in a public offering. In addition, the Company granted the Underwriter the option, for 45 days from the closing of the offering, to purchase up to 28,500 additional shares of common stock and common warrants to purchase up to an additional 72,500 shares of common stock. The Underwriter agreed to purchase the shares from the Company pursuant to at a price of $15.62 per share.
In lieu of a purchase of common stock that would otherwise result in an investor’s beneficial ownership exceeding 4.99% (or, at the election of the investor, 9.99%) of the outstanding common stock, a pre-funded warrant was offered, each of which enables the investor to purchase one share of common stock at an exercise price of $0.0001. Each pre-funded warrant was exercisable upon issuance and will expire when exercised in full (all pre-funded warrants were exercised immediately upon issuance). Each pre-funded warrant was sold with a common warrant to purchase two shares of common stock. The public purchase price of one share of common stock and accompanying common warrant to purchase two shares of Common Stock is $16.80 and the combined purchase price of one pre-funded warrant and accompanying common warrant to purchase two shares of common stock is $16.80.
Each common warrant is exercisable immediately at an exercise price of $16.80 per share and will expire five years following the date of issuance. The offering closed on December 9, 2022 and we received aggregate gross proceeds of approximately $4.1 million from the Offering.
2023 February Offering
On February 2, 2023, we entered into a definitive Securities Purchase Agreement with certain institutional investors , pursuant to which the Company agreed to issue and sell in a registered direct offering, priced “at-the-market” under the rules of The Nasdaq Stock Market, an aggregate of 297,619 shares of common stock of the Company, par value $0.0001 per share, at an offering price of $10.08 per share, for gross proceeds of approximately $3.0 million before the deduction of placement agent fees and related costs of $0.3 million. The closing of the Offering occurred on February 6, 2023.
| 71 |
In a concurrent private placement, the Company issued to the institutional investors, for each share of common stock purchased in the offering, a common warrant to purchase one share of common stock. The common warrants are exercisable immediately upon issuance and terminate five and one-half years following issuance. The common warrants have an exercise price of $8.58 per share and are exercisable to purchase an aggregate of up to 297,619 shares of Common Stock and expire on August 7, 2028. A holder of a common warrant will not have the right to exercise any portion of its warrants if the holder, together with its affiliates, would beneficially own in excess of 4.99% (or 9.99% at the election of the holder prior to the date of issuance) of the number of shares of common stock outstanding immediately after giving effect to such exercise; provided, however, that upon 61 days’ prior notice to the Company, the holder may increase or decrease the beneficial ownership limitation, provided that in no event shall the beneficial ownership limitation exceed 9.99%.
H.C. Wainwright & Co. acted as the exclusive placement agent (the “Placement Agent”) for the offering. We issued placement agent warrants to purchase up to 20,832 shares of common stock to the Placement Agent (including its designees). These warrants have an exercise price equal to $12.60 per share and are exercisable for five years from the commencement of sales in the offering. The common warrants and placement agent warrants and the shares of our common stock issuable upon the exercise of the common warrants and placement agent warrants are not being registered under the Securities Act of 1933, as amended, are not being offered pursuant to the Registration Statement, and are being offered pursuant to the exemption provided in Section 4(a)(2) under the Securities Act of 1933 and Rule 506(b).
In the Securities Purchase Agreement, we agreed not to issue, enter into any agreement to issue or announce the issuance or proposed issuance of any shares of common stock or any securities convertible into or exercisable or exchangeable for Common Stock for a period of 30 days following the closing of the offering. Our officers and directors agreed, subject to limited exceptions, for a period of 90 days after the closing of the offering, to not offer, sell, contract to sell, hypothecate, pledge or otherwise dispose of, directly or indirectly, or establish or increase a put equivalent position or liquidate or decrease a call equivalent position within the meaning of Section 16 of the Securities Exchange Act of 1934, as amended, with respect to, any shares of common stock or securities convertible, exchangeable or exercisable into, shares of common stock beneficially owned, held or thereafter acquired by them.
2023 May Offering
On May 12, 2023, the Company completed a public offering of an aggregate of 1,800,876 shares of its common stock at par value $0.0001 per share (including pre-funded warrants in lieu thereof), Series A-1 warrants to purchase up to 1,800,876 shares of common stock and Series A-2 warrants to purchase up to 1,800,876 shares of common stock, at a combined public offering price of $3.887 per share (or pre-funded warrant in lieu thereof) and accompanying warrants. The Series A-1 warrants have an exercise price of $3.64 per share, are exercisable immediately upon issuance and expire five years from the date of issuance, and the Series A-2 warrants have an exercise price of $3.64 per share, are exercisable immediately upon issuance and expire eighteen months from the date of issuance. A holder of a warrant issued in the offering will not have the right to exercise any portion of its warrants if the holder, together with its affiliates, would beneficially own in excess of 4.99% (or 9.99% at the election of the holder prior to the date of issuance) of the number of shares of Common Stock outstanding immediately after giving effect to such exercise; provided, however, that upon 61 days’ prior notice to the Company, the holder may increase or decrease the beneficial ownership limitation, provided that in no event shall the beneficial ownership limitation exceed 9.99%. Gross proceeds from this offering are approximately $7.0 million before the deduction of placement agent fees and related costs of $0.7 million. The Series A-1 and Series A-2 warrants were repriced to $1.31 per share and exercised in February 2024.
H.C. Wainwright & Co. acted as the exclusive placement agent for the offering. The Company also registered warrants issued to the placement agent to purchase 126,061 shares of common stock at a per share exercise price of $4.8588, which is 125% of the price of the shares in the offering.
In connection with the offering, the Company amended certain existing warrants to purchase up to an aggregate of 210,085 shares of the Company’s common stock that were previously issued in September 2021 through December 2022 to purchasers in the offering at exercise prices ranging from $16.80 to $187.20 per share, such that the amended warrants have a reduced exercise price of $3.64 per share, at an additional offering price of $0.125 per amended warrant.
| 72 |
2023 Notes
On October 23, 2023, the Company entered into a Securities Purchase Agreement (the “SPA”) for an aggregate financing of $1.7 million with investors, including $0.2 million with a board member. At the first closing under the SPA, which occurred on October 25, 2023, the Company issued to the investors (i) senior secured convertible promissory notes in the aggregate principal amount of $612,000 for an aggregate purchase price of $566,667 and (ii) warrants to purchase 1,255,697 shares of the Company’s common stock in the aggregate. At the second closing under the SPA, which occurred on November 28, 2023, the Company issued to the investors referenced above, (i) additional notes in the aggregate principal amount of $1,224,000 for an aggregate purchase price of $1,133,333 and (i) additional warrants to purchase 2,511,394 shares of the common stock in the aggregate. The notes mature on April 25, 2024 and May 28, 2024, respectively.
The combined notes are subject to an original issue discount of 8%, have a term of six months from their respective date of issuance and accrue interest at the rate of 6.0% per annum. The notes are convertible into common stock, at a per share conversion price equal to $1.5675. Beginning ninety days following issuance of the notes at the first closing and second closing, respectively, the Company is obligated to redeem monthly one third of the original principal amount under the applicable note, plus accrued but unpaid interest, liquidated damages and any other amounts then owing to the holder of such note. The Company is required to pay the redemption amount in cash with a premium of 10% or, at the election of the investor at any time, some or all of the principal amount and interest may be paid by conversion of shares under the note into common stock based on a conversion price equal to $1.5675. Conversions and repayments of principal and interest on the notes in January and February 2024 totaled $1.7 million.
The warrants have an exercise price of $1.5675 and are exercisable for five years following issuance on each of the first and second closing dates under the SPA. Warrants for 1.3 million shares of common stock were exercised in January 2024.
Components of Our Operating Results
Revenue
We have generated limited revenue since our inception and we do not expect to generate any revenue from the sale of products in the near future, if at all. If our development efforts are successful and we commercialize our products, or if we enter into collaboration or license agreements with third parties, we may generate revenue in the future from product sales, as well as upfront, milestone and royalty payments from such collaboration or license agreements, or a combination thereof.
We have received funding under federal grants from the National Institutes of Health (“NIH”) through the National Institute on Drug Abuse (“NIDA”). In September 2018, we were awarded a research and development grant related to the development of our MPAR® overdose prevention technology (the “MPAR Grant”). In September 2019, we were awarded a second research and development grant related to the development of our TAAP/MPAR® abuse deterrent technology for Opioid Use Disorder (“OUD”) (the “OUD Grant”). Grant funds are awarded annually through a Notice of Award which contains certain terms and conditions including, but not limited to, complying with the grant program legislation, regulation and policy requirements, complying with conditions on expenditures of funds with respect to other applicable statutory requirements such as the federal appropriations acts, periodic reporting requirements, and budget requirements.
| 73 |
Operating Expenses
Research and Development Expenses
Research and development expenses consist primarily of costs incurred for research activities, including drug discovery efforts and the development of our product candidates. We expense research and development costs as incurred, which include:
| ● | expenses incurred to conduct the necessary preclinical studies and clinical trials required to obtain regulatory approval; | |
| ● | expenses incurred under agreements with contract research organizations (“CROs”) that are primarily engaged in the oversight and conduct of our drug discovery efforts and preclinical studies, clinical trials and contract manufacturing organizations (“CMOs”) that are primarily engaged to provide preclinical and clinical drug substance and product for our research and development programs; | |
| ● | other costs related to acquiring and manufacturing materials in connection with our drug discovery efforts and preclinical studies and clinical trial materials, including manufacturing validation batches, as well as investigative sites and consultants that conduct our clinical trials, preclinical studies and other scientific development services; | |
| ● | payments made in cash or equity securities under third-party licensing, acquisition and option agreements; | |
| ● | employee-related expenses, including salaries and benefits, travel and stock-based compensation expense for employees engaged in research and development functions; | |
| ● | costs related to compliance with regulatory requirements; and | |
| ● | allocated facilities-related costs, depreciation and other expenses, which include rent and utilities. |
We recognize external development costs as incurred. Any advance payments that we make for goods or services to be received in the future for use in research and development activities are recorded as prepaid expenses. Such amounts are expensed as the related goods are delivered or the related services are performed, or until it is no longer expected that the goods will be delivered or the services rendered. We estimate and accrue for the value of goods and services received from CROs and other third parties each reporting period based on an evaluation of the progress to completion of specific tasks using information provided to us by our service providers. This process involves reviewing open contracts and purchase orders, communicating with our personnel to identify services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of actual costs.
We do not track our research and development expenses on a program-by-program basis. Our direct external research and development expenses consist primarily of external costs, such as fees paid to outside consultants, CROs, CMOs and research laboratories in connection with our preclinical development, process development, manufacturing and clinical development activities. We do not allocate employee costs, costs associated with our discovery efforts, laboratory supplies, and facilities, including depreciation or other indirect costs, to specific programs because these costs are deployed across multiple programs and, as such, are not separately classified. We use internal resources primarily to conduct our research and development as well as for managing our preclinical development, process development, manufacturing and clinical development activities. These employees work across multiple programs and, therefore, we do not track our costs by program and cannot state precisely the total costs incurred for each of our clinical and preclinical programs on a project-by-project basis.
| 74 |
Research and development activities are central to our business model. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. As a result, we expect that our research and development expenses will remain elevated as we continue our existing, and commences additional, planned clinical trials for PF614, PF614-MPAR® and nafamostat, as well as conduct other preclinical and clinical development, including submitting regulatory filings for our other product candidates, subject to our ability to obtain financing. We also expect our related personnel costs to increase and, as a result, we expect our research and development expenses, including costs associated with stock-based compensation, to remain elevated. In addition, we may incur additional expenses related to milestone and royalty payments payable to third parties with whom we may enter into license, acquisition and option agreements to acquire the rights to future product candidates.
At this time, we cannot reasonably estimate or know the nature, timing and costs of the efforts that will be necessary to complete the preclinical and clinical development of any of our product candidates or when, if ever, material net cash inflows may commence from any of our product candidates. The successful development and commercialization of our product candidates are highly uncertain. This uncertainty is due to the numerous risks and uncertainties associated with product development and commercialization, including the uncertainty of the following:
| ● | the scope, progress, outcome and costs of our preclinical development activities, clinical trials and other research and development activities; | |
| ● | establishing an appropriate safety and efficacy profile with investigational new drug (“IND”) enabling studies; | |
| ● | successful patient enrollment in and the initiation and completion of clinical trials; | |
| ● | the timing, receipt and terms of any marketing approvals from applicable regulatory authorities including the FDA and non-U.S. regulators; | |
| ● | the extent of any required post-marketing approval commitments to applicable regulatory authorities; | |
| ● | establishing clinical and commercial manufacturing capabilities or making arrangements with third-party manufacturers in order to ensure that we or our third-party manufacturers are able to make product successfully; | |
| ● | development and timely delivery of clinical-grade and commercial-grade drug formulations that can be used in our clinical trials and for commercial launch; | |
| ● | obtaining, maintaining, defending and enforcing patent claims and other intellectual property rights; | |
| ● | significant and changing government regulation; | |
| ● | launching commercial sales of our product candidates, if and when approved, whether alone or in collaboration with others; and | |
| ● | maintaining a continued acceptable safety profile of our product candidates following approval, if any, of our product candidates. |
Any changes in the outcome of any of these variables with respect to the development of our product candidates in preclinical and clinical development could mean a significant change in the costs and timing associated with the development of these product candidates. For example, if the FDA or another regulatory authority were to delay our planned start of clinical trials or require us to conduct clinical trials or other testing beyond those that we currently expect or if we experience significant delays in enrollment in any of our planned clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development of that product candidate.
| 75 |
General and Administrative Expenses
General and administrative expenses consist primarily of employee-related expenses, including salaries and related benefits, travel and stock-based compensation for personnel in executive, business development, finance, human resources, legal, information technology, and administrative functions. General and administrative expenses also include direct and allocated facility-related costs as well as insurance costs and professional fees for legal, patent, consulting, investor and public relations, accounting and audit services. We expense general and administrative costs as incurred.
We anticipate that our general and administrative expenses will increase in the future as we increase our headcount to support the continued development of our product candidates, subject to our ability to obtain financing. We also anticipate that we will continue to incur significant accounting, audit, legal, regulatory, compliance and director and officer insurance costs as well as investor and public relations expenses as a public company. Additionally, if and when we believe a regulatory approval of a product candidate appears likely, we anticipate an increase in payroll and other employee-related expenses as a result of our preparation for commercial operations, especially as it relates to the sales and marketing of that product candidate.
Other Income (Expense)
Loss on issuance of convertible notes
The 2022 Notes were accounted for under ASC 480 – Distinguishing Liabilities from Equity, due to share settlement features contained within the notes. As a result, the 2022 Notes were recorded as liabilities at fair value upon initial recognition and at the balance sheet date. We used a discounted cash flow model and a Monte Carlo simulation to estimate the fair value of the notes, both of which rely on unobservable Level 3 inputs. The loss on issuance of convertible notes represents the difference between the gross proceeds received and the calculated fair value on the issuance date of the notes.
Issuance costs for convertible notes
The issuance costs for convertible notes represent the original issue discount (expensed immediately due to the initial recognition at fair value of the 2022 Notes noted above), and legal and accounting fees incurred in connection with the issuance of the 2022 Notes.
Loss on conversions and change in fair value of convertible notes
When conversions on the 2021 Notes occurred, we calculated the difference between the conversion price and the average of the high and low stock price on the date of conversion. The resulting difference was either a loss if the conversion price was below the average of the high and low stock price on the date of conversion or a gain if the conversion price was above the average of the high and low stock price on the date of conversion.
We elected the fair value option to account for the 2021 Notes as we believe the fair value option provided users of the financial statements with greater ability to estimate the outcome of future events as facts and circumstances change, particularly with respect to changes in the fair value of the common stock underlying the conversion option. The 2022 Notes were accounted for under ASC 480 – Distinguishing Liabilities from Equity, due to share settlement features contained within the notes. We used a discounted cash flow model and a Monte Carlo simulation to estimate the fair value of the notes, both of which rely on unobservable Level 3 inputs. Changes in the fair value of the notes are recognized through earnings for each reporting period.
Issuance of liability classified warrants
The warrants issued with the 2021 Notes and 2022 Notes are liability classified due to certain cash settlement features. We use a Black-Scholes option pricing model to estimate the fair value of the warrants. This represents the immediate expense upon initial recognition of the liability that is included in the statement of operations. The liability is remeasured each reporting period as described further below.
| 76 |
Change in fair value of liability classified warrants
We use a Black-Scholes option pricing model to estimate the fair value of the liability classified warrants. Changes in the fair value of the warrants are recognized through earnings for each reporting period.
Interest Expense
Interest expense consists of interest accrued on our financed directors’ and officers’ insurance, and accumulated interest from the 2023 Notes based on the stated interest rate. In addition, the 2023 Notes balance at December 31, 2023 reflects amortization of the debt discount from the original issuance and a discount associated with the warrant issuances and amortization of the associated debt issuance costs that are all recorded as interest expense. Interest expense related to the 2021 Notes and 2022 Notes was included in the estimate of fair value of the convertible notes.
Provision for Income Taxes
We have not recorded any significant amounts related to income tax expense, we have not recognized any reserves related to uncertain tax positions, nor have we recorded any income tax benefits for the majority of our net losses we have incurred to date or for our research and development tax credits.
We account for income taxes using the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements or our tax returns. Deferred tax assets and liabilities are determined based on difference between the financial statement carrying amounts and tax bases of existing assets and liabilities and for loss and credit carryforwards, which are measured using the enacted tax rates and laws in effect in the years in which the differences are expected to reverse. The realization of our deferred tax assets is dependent upon the generation of future taxable income, the amount and timing of which are uncertain. Valuation allowances are provided, if, based upon the weight of available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized. As of December 31, 2023 and 2022, we continue to maintain a full valuation allowance against all of our deferred tax assets based on our evaluation of all available evidence.
Beginning in 2022, the Tax Cuts and Jobs Act, or the Tax Act, eliminated the option to deduct research and development expenditures currently and requires taxpayers to capitalize and amortize them over five or fifteen years pursuant to Internal Revenue Code Section 174. This has not impacted our effective tax rate or our cash tax payable in 2023; however, if the requirement to capitalize Section 174 expenditures is not modified, it may also impact our effective tax rate and our cash tax liability in future years.
We file income tax returns in the United States federal tax jurisdiction and state jurisdictions and may become subject to income tax audit and adjustments by related tax authorities. Our tax return period for United States federal income taxes for the tax years since 2020 remain open to examination under the statute of limitations by the Internal Revenue Service and state jurisdictions are open for examination from 2019. We record reserves for potential tax payments to various tax authorities related to uncertain tax positions, if any. The nature of uncertain tax positions is subject to significant judgment by management and subject to change, which may be substantial. These reserves are based on a determination of whether and how much a tax benefit taken by us in our tax filings or whether our position is more likely than not to be realized following the resolution of any potential contingencies related to the tax benefit. We develop our assessment of uncertain tax positions, and the associated cumulative probabilities, using internal expertise and assistance from third-party experts. As additional information becomes available, estimates are revised and refined. Differences between estimates and final settlement may occur resulting in additional tax expense. Potential interest and penalties associated with such uncertain tax positions is recorded as a component of our provision for income taxes. To date, no amounts are being presented as an uncertain tax position.
The following table summarizes our results of operations for the years ended December 31, 2023 and 2022:
| 77 |
Results of Operations
Comparison of the Years ended December 31, 2023 and 2022
| Year Ended December 31, | ||||||||||||
| 2023 | 2022 | Change | ||||||||||
| Federal grants | $ | 2,230,520 | $ | 2,523,383 | $ | (292,863 | ) | |||||
| Operating expenses: | ||||||||||||
| Research and development | $ | 7,587,473 | $ | 19,835,875 | $ | (12,248,402 | ) | |||||
| General and administrative | 5,361,234 | 6,909,603 | (1,548,369 | ) | ||||||||
| Total operating expenses | 12,948,707 | 26,745,478 | (13,796,771 | ) | ||||||||
| Loss from operations | (10,718,187 | ) | (24,222,095 | ) | 13,503,908 | |||||||
| Other income (expense): | ||||||||||||
| Loss on issuance of convertible notes | - | (3,609,944 | ) | 3,609,944 | ||||||||
| Issuance costs for convertible notes | - | (1,137,740 | ) | 1,137,740 | ||||||||
| Loss on conversions and change in fair value of convertible notes | 146,479 | 1,792,154 | (1,645,675 | ) | ||||||||
| Issuance of liability classified warrants | - | (3,737,371 | ) | 3,737,371 | ||||||||
| Change in fair value of liability classified warrants | 283,958 | 6,730,613 | (6,446,655 | ) | ||||||||
| Interest expense | (353,945 | ) | (109,525 | ) | (244,420 | ) | ||||||
| Other income and expense, net | 15,420 | 86,223 | (70,803 | ) | ||||||||
| Total other income/(expenses), net | 91,912 | 14,410 | 77,502 | |||||||||
| Net loss | $ | (10,626,275 | ) | $ | (24,207,685 | ) | $ | 13,581,410 | ||||
| Net loss attributable to noncontrolling interests | (13,201 | ) | (35,393 | ) | 22,192 | |||||||
| Deemed dividend related to warrants down round provision | 12,937 | 913,204 | (900,267 | ) | ||||||||
| Net loss attributable to common stockholders | $ | (10,626,011 | ) | $ | (25,085,496 | ) | $ | 14,459,485 | ||||
Federal Grants
Revenue from federal grants totaled $2.2 million for the year ended December 31, 2023, compared to $2.5 million for the year ended December 31, 2022. Revenue decreased $0.3 million during the year ended December 31, 2023, due to the timing of research activities eligible for funding under the grants.
Research and Development Expenses
Research and development expenses were $7.6 million for the year ended December 31, 2023, compared to $19.8 million for the year ended December 31, 2022. The decrease was primarily the result of changes in timing of external research and development costs related to the clinical programs for PF614 and PF614-MPAR. We do not currently track expenses on a program-by-program basis. We expect future research and development expenses to approximate current levels but may need to be adjusted based on our ability to raise capital sufficient to fund these expenses.
General and Administrative Expenses
General and administrative expenses were $5.4 million for the year ended December 31, 2023, compared to $6.9 million for the year ended December 31, 2022. The decrease was primarily a result of reduced stock-based compensation, reduced liability insurance, legal, and consulting fees and no employee bonus expenses in the 2023 period. We expect future general and administrative expenses to approximate current levels.
| 78 |
Other Income and Expense
Other income and expenses, net, were $91,912 for the year ended December 31, 2023, compared to $14,410 for the year ended December 31, 2022. The decrease was primarily the result of interest expense in the current year offset by changes in the fair value of certain financial instruments. The activity from 2022 included the accounting considerations of the conversions of the 2021 Notes as well as the issuance of the 2022 Notes. There was no corresponding conversions activity in the 2023 period associated with the 2022 Notes due to the accounting under ASC 480. Interest expense incurred in 2023 is primarily driven by the amortization of debt discount and debt issuance costs related to the 2023 Notes.
Liquidity and Capital Resources
Sources of Liquidity and Capital
As of December 31, 2023, we had $1.1 million of cash and cash equivalents. Since inception, we have generated limited revenues and have incurred significant operating losses and negative cash flows from our operations, and we anticipate that we will continue to incur losses for at least the foreseeable future. We have not yet commercialized any of our product candidates and we do not expect to generate revenue from sales of any product candidates for several years, if at all.
We have funded our operations to date primarily with proceeds from the sale of common equity, funding under federal research grants and borrowings under convertible promissory notes. To fund future operations, we will need to raise additional capital. The amount and timing of future funding requirements will depend on many factors, including the timing and results of our ongoing research and development efforts and related general and administrative support. We anticipate that we will fund our operations through public or private equity or debt financings or other sources, such as potential collaboration agreements. We cannot make assurances that anticipated additional financing will be available to us on favorable terms, if at all.
Remaining funding under approved federal research grants totals $2.2 million and is expected to be utilized by August 2024. Pursuant to the terms and conditions of the two grants, we are required to submit progress reports to NIDA on an annual basis and a final research performance progress report within 120 days of the performance period end date. Additionally, the grants limit the use of funds to activities that are clearly severable and independent from activities that involve human subjects until the receipt by NIDA of (i) Institutional Review Board (“IRB”) approval, (ii) federal-wide assurance from the Office for Human Research Protections, (iii) a Data and Safety Monitoring Plan, (iv) certification that all key personnel have completed education on the protection of human subjects and (v) a Clinical Trials Dissemination Plan. We must also comply with the data sharing policies of NIDA and the NIH Public Access Policy, that require submission of final peer-reviewed journal manuscripts that arise from the use of grants to PubMed Central immediately upon acceptance for publication.
Neither grant must be repaid. To receive the remaining funding for each respective study covered by a grant, we must meet certain milestones. We have met the required milestones under the MPAR Grant. The remaining milestone under the OUD Grant is identification of a R-methadone-TAAP clinical candidate that meet the specified criteria.
Inventions arising from the research projects funded with the grants are required to be reported to NIDA, per the Bayh-Dole Act (the Patent and Trademark Law Amendments Act), that permits us to retain ownership of the inventions, while also giving NIDA the license to practice the subject invention. In turn, we are expected to file for patent protection and to ensure commercialization upon licensing for the benefit of public health.
| 79 |
Pursuant to the GEM Agreement, we are entitled to draw down up to $60.0 million of gross proceeds (“Aggregate Limit”) from GEM Global in exchange for shares of our common stock, subject to meeting the terms and conditions of the GEM Agreement. This share subscription facility is available for a period of 36 months from the closing date of the Merger (July 2024). A draw down is subject to limitations on the amount that is drawn under the facility and must comply with certain conditions precedent including the listing of our shares on a principal market (which includes Nasdaq), having the necessary number of shares that are issuable pursuant to the draw down registered under an effective registration statement, and other notice and timing requirements. Upon our valid exercise of a draw down, pursuant to delivery of a notice and in accordance with other conditions, GEM Global is required to pay, in cash, a per-share amount equal to 90% of the average closing bid price of the shares of our common stock recorded by Nasdaq during the 30 consecutive trading days commencing on the first trading day that is designated on the draw down notice. In no event may our draw down requests exceed 400% (“Draw Down Limit”) of the average daily trading volume for the 30 trading days immediately preceding the date we deliver the draw down notice. We may not be able to utilize the facility before it expires. Our ability to utilize this share subscription facility is restricted while financing commitments to which we are subject remain outstanding.
Upon the public listing of the Company’s shares following the closing of the Merger, GEM Global became entitled to a commitment fee in the form of cash or freely tradeable shares of our common stock in an amount equal to 2% of the Aggregate Limit or $1.2 million to be paid in two tranches. The commitment fee for the first tranche, which was equal to 67% of the commitment fee, or $800,000, was discharged with 3,838 shares of common stock transferred from related parties in July 2022. The commitment fee for the second tranche, which was equal to the remaining 33% of the commitment fee, or $400,000 was paid in January 2023 through the issuance of 44,444 shares of registered common stock.
Additionally, we issued a warrant with a 36-month term at the closing of the Merger granting GEM Global the right to purchase 4,608 shares of our common stock (an amount equal to 4% of the total number of our common stock outstanding as of the closing date of the Merger (subject to adjustments described below), calculated on a fully diluted basis), at a strike price per share equal to $2,402.40, which was the closing bid price for such common stock on the first day of trading on Nasdaq. The exercise price was reduced to $1.5675 per share as of December 31, 2023 because of a pricing adjustment per the GEM Agreement which is reflected on the consolidated statement of operations as a deemed dividend. The warrant can be exercised on a cashless basis in part or in whole at any time during the term. Any failure by us to timely transfer the shares under the warrant pursuant to GEM Global’s exercise will entitle GEM Global to compensation in addition to other remedies. The number of shares underlying the warrant as well as the strike price is subject to adjustments for recapitalizations, reorganizations, change of control, stock split, stock dividend, reverse stock splits, and issuances of additional common shares at a price per share less than the exercise price.
Pursuant to the terms of the GEM Agreement, we are required to indemnify GEM Global for any losses it incurs as a result of a breach by us or of our representations and warranties and covenants under the GEM Agreement or for any misstatement or omission of a material fact in a registration statement registering those shares pursuant to the GEM Agreement. Also, GEM Global is entitled to be reimbursed for legal or other costs or expenses reasonably incurred in investigating, preparing, or defending against any such loss.
Going Concern
We have generated limited revenues and have incurred significant operating losses since our inception and, as of December 31, 2023, we have an accumulated deficit of $121.6 million. We expect to continue to incur significant expenses and operating losses for the foreseeable future. Without capital raised through financing transactions, existing cash resources are sufficient to allow us to fund current planned operations into the third quarter of 2024, which raises substantial doubt about the Company’s ability to continue as a going concern.
For additional information on risks associated with our substantial capital requirements, please read the section titled “Risk Factors” included elsewhere in this Annual Report on Form 10-K.
| 80 |
Cash Flows for the years ended December 31, 2023 and 2022
The following table summarizes our cash flows for each of the periods presented:
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Net cash used in operating activities | $ | (10,779,982 | ) | $ | (17,887,439 | ) | ||
| Net cash provided by investing activities | - | 4,500 | ||||||
| Net cash provided by financing activities | 8,755,884 | 8,765,905 | ||||||
| Net decrease in cash and cash equivalents | $ | (2,024,098 | ) | $ | (9,117,034 | ) | ||
Operating Activities
During the years ended December 31, 2023 and 2022, we used cash in operating activities of $10.8 million and $17.9 million, respectively. The decrease primarily resulted from the change in net loss between 2022 and 2023 and the timing of vendor invoicing and payments primarily associated with research and development activities.
Financing Activities
During the years ended December 31, 2023 and 2022, net cash provided by financing activities was $8.7 million and $8.8 million, respectively. For 2023, net cash proceeds from 2023 February and 2023 May offerings of $9.1 million, net of transaction costs of $0.4 million, net cash proceeds from 2023 Notes of $1.6 million, and the repayment of financed insurance premiums of $0.5 million and cash payment of 2022 Notes of $1.0 million. In 2022, net cash consisted primarily of net proceeds from the issuance of the 2022 Notes and the net proceeds of the issuance of shares and related warrants in connection with the underwriting agreement completed in December of 2022.
Funding Requirements
Our primary use of cash is to fund operating expenses, primarily related to our research and development activities. Cash used to fund operating expenses is impacted by the timing of when we pay these expenses, as reflected in the change in our outstanding accounts payable, accrued expenses and prepaid expenses.
We expect our expenses, excluding non-cash expenses to recognize the fair value of warrants and convertible notes, to remain elevated in connection with our ongoing activities, particularly as we advance the preclinical activities and clinical trials of our product candidates. In addition, upon the completion of the Merger, we have incurred, and will continue to incur, additional costs associated with operating as a public company, including significant legal, accounting, insurance, investor relations and other expenses. The timing and amount of our operating expenditures will depend largely on our ability to:
| ● | advance preclinical development of our early-stage programs and clinical trials of our product candidates; | |
| ● | manufacture, or have manufactured on our behalf, our preclinical and clinical drug material and develop processes for late state and commercial manufacturing; | |
| ● | seek regulatory approvals for any product candidates that successfully complete clinical trials; | |
| ● | establish a sales, marketing, medical affairs and distribution infrastructure to commercialize any product candidates for which we may obtain marketing approval and intend to commercialize on our own; | |
| ● | hire additional clinical, quality control and scientific personnel; |
| 81 |
| ● | expand our operational, financial and management systems and increase personnel, including personnel to support our clinical development, manufacturing and commercialization efforts and our operations as a public company; | |
| ● | obtain, maintain, expand and protect our intellectual property portfolio; | |
| ● | manage the costs of preparing, filing and prosecuting patent applications, maintaining and protecting our intellectual property rights, including enforcing and defending intellectual property related claims; and | |
| ● | manage the costs of operating as a public company. |
Commitments
Our commitments as of December 31, 2023 included an estimated $17.9 million related to open purchase orders and contractual obligations that occurred in the ordinary course of business, including commitments with contract research organizations for multi-year pre-clinical and clinical research studies. Although open purchase orders are considered enforceable and legally binding, the terms generally allow us the option to cancel, reschedule, and adjust requirements based on our business needs prior to the delivery of goods or the performance of services.
Working Capital
Because of the numerous risks and uncertainties associated with research, development and commercialization of biologic product candidates, we are unable to estimate the exact amount of our working capital requirements. Our future funding requirements will depend on and could increase significantly as a result of many factors, including:
| ● | the scope, progress, results and costs of researching and developing our product candidates, and conducting preclinical and clinical trials; | |
| ● | the costs, timing and outcome of regulatory review of our product candidates; | |
| ● | the costs, timing and ability to manufacture our product candidates to supply our clinical and preclinical development efforts and our clinical trials; | |
| ● | the costs of future activities, including product sales, medical affairs, marketing, manufacturing and distribution, for any of our product candidates for which we receive marketing approval; | |
| ● | the costs of manufacturing commercial-grade product and necessary inventory to support commercial launch; | |
| ● | the ability to receive additional non-dilutive funding, including grants from organizations and foundations; | |
| ● | the revenue, if any, received from commercial sale of our products, should any of our product candidates receive marketing approval; | |
| ● | the costs of preparing, filing and prosecuting patent applications, obtaining, maintaining, expanding and enforcing our intellectual property rights and defending intellectual property-related claims; | |
| ● | our ability to establish and maintain collaborations on favorable terms, if at all; and | |
| ● | the extent to which we acquire or in-license other product candidates and technologies. |
| 82 |
Critical Accounting Policies and Significant Judgments and Estimates
Our consolidated financial statements are prepared in accordance with GAAP. The preparation of our consolidated financial statements and related disclosures requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, costs and expenses. We base our estimates on historical experience, known trends and events and various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. We evaluate our estimates and assumptions on an ongoing basis. Our actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in Note 3 to our audited consolidated financial statements, we believe that the following accounting policy is the most critical to the judgments and estimates used in the preparation of our consolidated financial statements.
Accrued Research and Development Expenses
As part of the process of preparing our consolidated financial statements, we are required to estimate our accrued research and development expenses. This process involves reviewing open contracts and purchase orders, communicating with our applicable personnel to identify services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when it has not yet been invoiced or otherwise notified of actual costs. Many of our service providers invoice us in arrears for services performed, on a pre-determined schedule or when contractual milestones are met; however, some require advance payments. We make estimates of our accrued expenses as of each balance sheet date in the consolidated financial statements based on facts and circumstances known to us at that time. We periodically confirm the accuracy of the estimates with the service providers and adjust if necessary. Examples of estimated accrued research and development expenses include fees paid to:
| ● | vendors, including research laboratories, in connection with preclinical development activities; | |
| ● | CROs and investigative sites in connection with preclinical studies and clinical trials; and | |
| ● | CMOs in connection with drug substance and drug product formulation of preclinical studies and clinical trial materials. |
We base our expenses related to preclinical studies and clinical trials on our estimates of the services received and efforts expended pursuant to quotes and contracts with multiple research institutions and CROs that supply, conduct and manage preclinical studies and clinical trials on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. There may be instances in which payments made to our vendors will exceed the level of services provided and result in a prepayment of the expense. Payments under some of these contracts depend on factors such as the successful enrollment of patients and the completion of clinical trial milestones. In accruing service fees, we estimate the time period over which services will be performed and the level of effort to be expended in each period. If the actual timing of the performance of services or the level of effort varies from the estimate, we adjust the accrual or the prepaid expense accordingly. Although we do not expect our estimates to be materially different from amounts actually incurred, our understanding of the status and timing of services performed relative to the actual status and timing of services performed may vary and may result in reporting amounts that are too high or too low in any period.
Off-Balance Sheet Arrangements
We do not have during the periods presented, and do not currently have, any off-balance sheet arrangements, as defined in the rules and regulations of the SEC.
| 83 |
Recently Issued Accounting Pronouncements
A description of recently issued accounting pronouncements that may potentially impact our financial position and results of operations is disclosed in Note 3 to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
Smaller Reporting Company Status
We are a “smaller reporting company” as defined in Item 10(f)(1) of Regulation S-K. Smaller reporting companies may take advantage of certain reduced disclosure obligations, including, among other things, providing only two years of audited financial statements. We will remain a smaller reporting company until the last day of the fiscal year in which (i) the market value of our common stock held by non-affiliates exceeds $250 million as of the prior June 30, or (ii) our annual revenues exceeded $100 million during such completed fiscal year and the market value of our common stock held by non-affiliates exceeds $700 million as of the prior June 30.
Item 7A. Quantitative and Qualitative Disclosure About Market Risk
We are exposed to market risk in the ordinary course of our business. These risks primarily relate to changes in interest rates and inflation.
Interest Rate Risk
Our cash and cash equivalents as of December 31, 2023, consisted of cash and a money market fund account. Because of the short-term nature of our money market fund, a sudden change in market interest rates would not be expected to have a material impact on our financial position or results of operations.
Inflation Risk
We do not believe that inflation and changing prices had a significant impact on our results of operations for any periods presented herein.
Item 8. Financial Statements and Supplementary Data
The financial statements required by this Item are included in Item 15 of this report and are presented beginning on page F-1.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Our disclosure controls and procedures are designed to ensure that information required to be disclosed in the reports that we file or submit under the Securities Exchange Act of 1934 as amended (the “Exchange Act”) is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure. Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures (as defined in Exchange Act Rule 13a–15(e) and 15d-15(e)) as of December 31, 2023. Based on that evaluation, the Chief Executive Officer and Chief Financial Officer concluded that the Company’s disclosure controls and procedures were effective as of December 31, 2023.
| 84 |
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over our financial reporting. Internal control over financial reporting is defined in Rule 13a-15(f) under the Exchange Act as a process designed by, or under the supervision of, the company’s executive and financial officers and effected by the company’s board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes and includes those policies and procedures that (a) pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of the assets of the company; (b) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (c) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements, errors, or fraud. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
As of December 31, 2023, our management assessed the effectiveness of our internal control over financial reporting using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework (2013) (the “2013 Framework”). In adopting the 2013 Framework, management assessed the applicability of the principles within each component of internal control and determined whether they have been adequately addressed within the current system of internal control and adequately documented. Based on this assessment, management, under the supervision and with the participation of our Chief Executive Officer and Chief Financial Officer, concluded that, as of December 31, 2023, our internal control over financial reporting was effective based on these criteria.
This Annual Report on Form 10-K does not include an attestation report of our independent registered public accounting firm regarding internal control over financial reporting. As a smaller reporting company, we were not required to have, nor have we, engaged our independent registered public accounting firm to perform an audit of internal control over financial reporting pursuant to SEC rules that permit us to provide only management’s report in this Annual Report on Form 10-K.
Changes in Internal Control Over Financial Reporting
Management worked with third-party accounting consultants to develop a control structure that can be consistently applied to period-end financial reporting as well as to unusual and infrequent complex accounting transactions. Testing of these remediations was satisfactorily completed during the quarter ended December 31, 2023. There were no other changes in our internal control over financial reporting (as such term is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the quarter ended December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
Not applicable.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
Not applicable.
| 85 |
PART III
Item 10. Executive Officers and Directors
The following persons are our executive officers and directors:
| Name | Age* | Position | ||||
| Executive Officers | ||||||
| Lynn Kirkpatrick, Ph.D.** | 67 | President, Chief Executive Officer and Class III Director | ||||
| Geoffrey Birkett | 61 | Chief Commercial Officer | ||||
| David Humphrey | 55 | Chief Financial Officer, Secretary and Treasurer | ||||
| Jeffrey Millard, Ph.D. | 48 | Chief Operating Officer (consultant) | ||||
| Linda Pestano, Ph.D. | 55 | Chief Development Officer | ||||
| William Schmidt, Ph.D. | 73 | Chief Medical Officer | ||||
| Directors** | ||||||
| Andrew Benton, J.D. | 71 | Class I Director | ||||
| William Chang | 67 | Class I Director | ||||
| Bob Gower, Ph.D. | 86 | Class II Director and Chairman of the Board | ||||
| Adam S. Levin, M.D. | 45 | Class III Director | ||||
| Steve R. Martin | 62 | Class III Director | ||||
| Lee Rauch | 70 | Class I Director | ||||
| Curtis Rosebraugh, M.D., MPH | 66 | Class II Director | ||||
*Ages presented as of December 31, 2023
** Information about Dr. Kirkpatrick is set forth under “Executive Officers”
Information about our Executive Officers and Directors
Executive Officers
Lynn Kirkpatrick, Ph.D. has served as our Chief Executive Officer since January 2009. Dr. Kirkpatrick has spent over 30 years in drug discovery and development, has initiated the clinical development of four novel drug candidates and now strives to bring highly novel and safe pain therapies to commercialization. She received a Doctor of Philosophy (“Ph.D.”) degree in Medicinal and Biomedicinal Chemistry at the University of Saskatchewan, completed a Post-Doctoral Fellowship at the Yale University School of Medicine, and became a tenured full professor in the Department of Chemistry at the University of Regina. She co-founded ProlX Pharmaceuticals, Corp. (“ProlX”) an oncology discovery company, becoming Chief Executive Officer and successfully bringing three small molecules from discovery into clinical development, two of these her own discoveries from academia. ProlX was acquired by Biomira Inc., and Dr. Kirkpatrick became the Chief Scientific Officer of the merged company to focus on the development of oncology products and vaccines. In 2009, she co-founded PHusis Therapeutics, developing targeted small molecule precision medicines for oncology. At the same time, she became our Chief Executive Officer. Dr. Kirkpatrick has published extensively in the area of targeted drug discovery, abuse deterrent pain products and holds numerous patents for novel drugs and modalities. We believe Dr. Kirkpatrick is qualified to serve on our Board because of her extensive executive experience in our industry and her service as our Chief Executive Officer.
Geoffrey Birkett has served as our Chief Commercial Officer since October 2018. He has over 30 years of experience in the Pharmaceutical and Biotechnology area. He started his career as a biochemist at the Royal Victoria Infirmary in Newcastle-upon-Tyne, England. He then moved into the pharmaceutical industry, where he focused on pain/addiction and neuroscience throughout his career. He has developed and launched several groundbreaking therapies, including Nicorette (POM) and (OTC), Lexapro and several other psychiatry agents with Lundbeck. Mr. Birkett assisted on the launch of Prozac and Humatrope (human growth hormone) with Eli Lilly. He assisted in moving Seroquel from Phase 2 to global market leader with multi-billion dollar sales and he also participated in the launch of Zomig for migraines, which became a European market leader. He worked for most of his pharmaceutical career at AstraZeneca plc in both the United Kingdom and the United States, where he held many roles including overseeing the global oncology division. When the AstraZeneca merger took place, Mr. Birkett ran the merger process outside the United States across all markets, and ran a corporate change program to streamline research and development involving 67,000 staff. Since leaving AstraZeneca, Mr. Birkett has held multiple roles in biotech companies as senior officer or as a consultant. He is co-founder of a novel drug delivery company and has consulted for IPSOS, a large global research and consulting firm. He also served as president for North America/Canada of INDIVIOR, a large company producing addiction treatment drugs. Mr. Birkett joined us in 2018 and is focused on building a world class commercial team. Mr. Birkett attended Henley Business College in London and INSEAD Business School in France where he studied general management and a global leadership.
| 86 |
David Humphrey has served as our Chief Financial Officer since February 2021. Prior to joining the Company, Mr. Humphrey was most recently Chief Financial Officer of Senomyx, Inc. (“Senomyx”), a publicly held biotechnology company focused on taste science. In his previous employment, he guided public company financial reporting, including Forms 10-K, 10-Q, 8-K, S-3, S-8, proxy statements and SOX internal controls compliance, and acted as primary liaison with the audit committee and external auditors. Mr. Humphrey advised Senomyx’s board of directors, as part of core executive management team, in a $75 million acquisition by Firmenich SA, a private Swiss multinational flavor and fragrance company. Previously, he held finance and accounting leadership positions and consulted at numerous life sciences companies, including ActivX Biosciences, Aurora Biosciences and Gensia. Mr. Humphrey started his career as an accountant at Price Waterhouse. He holds a Bachelor of Science with Honors in Accountancy from the University of Illinois at Urbana-Champaign and is a Certified Public Accountant (inactive) in California.
Jeffrey Millard, Ph.D. has served as our Chief Operating Officer since January 2019. Dr. Millard has both academic and industrial experience in chemistry and pharmaceutical sciences covering all aspects of chemistry, manufacturing, and controls, or CMC. He has been involved in both start-up biotech as well as small and mid-sized public biopharmaceutical companies. Dr. Millard has been directly responsible for research and development activities and writing of more than seven IND submissions and Investigational Medicinal Product Dossiers, or IMPDs. He has directed the CMC efforts from discovery and in-licensing through commercial launch activities. His experience covers the application programming interface, or API, lifecycle (from synthetic route scouting, process chemistry, analytical chemistry development and validation, cGMP production and release of API, to QbD and process validation), and drug product development through manufacture. Dr. Millard received a Bachelor of Arts from Rice University and a Ph.D. in Pharmaceutical Sciences from the University of Arizona.
Linda Pestano, Ph.D. joined Ensysce in October 2021, as Chief Development Officer. Dr. Pestano has worked throughout her career to guide the development of novel therapeutics to improve patient outcomes and quality of life. She has 20 years of experience developing vaccines, drugs and novel biologics for a diverse range of indications. She has been instrumental in guiding new therapies, including small molecules, nucleic acids, and biologicals through development into clinical trials. Dr. Pestano’s expertise spans lead development, pre-clinical and translational studies, and interacting with multiple regulatory agencies. Dr. Pestano received her PhD from Tufts University and undertook a Post-Doctoral Fellowship with Dana Farber Cancer Institute at the Harvard Medical School in Boston.
William K. Schmidt, Ph.D., has served as our Chief Medical Officer since January 2016. He is also the Head of NorthStar Consulting, the Parliamentarian and a former president of the Eastern Pain Association, the largest regional affiliate of the American Pain Society. He has over 25 years of pharmaceutical industry experience with a special emphasis on the discovery and development of novel analgesic and narcotic antagonist drugs. He was previously Vice President of Clinical Development for CrystalGenomics (Seoul, South Korea) and its United States subsidiary, CG Pharmaceuticals (Emeryville, CA); Senior Vice President of Development at Limerick BioPharma; Vice President, Clinical Research, for Renovis, Inc.; and Vice President, Scientific Affairs and acting Vice President, Clinical Research and Development, at Adolor Corporation. At Adolor Corporation, Dr. Schmidt was a key member of the team leading to the clinical development, NDA filing, and FDA approval of Entereg® (alvimopan), a peripherally acting opioid antagonist. Currently Dr. Schmidt serves as an expert on pain medicine pharmaceutical development with pharmaceutical and biotech companies throughout North America, Europe, Asia, Latin America, and Australia. Dr. Schmidt received a Bachelor of Arts degree from the University of California Berkeley and his Ph.D. University of California-San Francisco.
Directors
Andrew Benton, J.D. has served as a member of our Board since December 2, 2019. Mr. Benton was the President, Chief Executive Officer and Trustee of Pepperdine University from June 2000 to July 2019. Mr. Benton was the former chairman of both the American Council of Education, the major coordinating body for all of the nation’s higher education institutions, and the National Association of Independent Colleges and Universities. Mr. Benton is also past chair of the Association of Independent California Colleges and Universities and a member of the American Bar Association, the Council for Higher Education Accreditation, the President’s Cabinet of the West Coast Conference, the Association of Presidents of Independent Colleges and Universities, and the Los Angeles World Affairs Council. Mr. Benton holds an undergraduate degree in American studies from Oklahoma Christian University and a J.D. from Oklahoma University. We believe that Mr. Benton’s experience governing academic and other institutions qualifies him to serve on our Board.
| 87 |
William Chang serves as Chief Executive Officer of Westlake Realty Group and Chairman of Westlake International Group where he has worked for more than 40 years. Mr. Chang is an investor in the San Francisco Giants of Major League Baseball. Mr. Chang was the former Chairman of U.S. Rugby Football Union. He also served on the Board of the Asia Foundation and San Francisco Port and Social Services Commissions. Mr. Chang holds a Bachelor’s degree in Economics from Harvard University. We believe that Mr. Chang’s extensive business experience and expertise in corporate governance qualifies him to serve on our Board.
Bob Gower, Ph.D. has served as our Chairman since 2008. Dr. Gower was Chief Executive Officer of Lyondell Petrochemical from 1985 through his retirement at the end of 1996. In 1997, he acquired businesses from Howell Corporation that became Specified Fuels and Chemicals. Dr. Gower sold Specified in 2000 and, with Dr. Richard Smalley, founded Carbon Nanotechnologies, Inc. (“CNI”) that same year to develop production capabilities and applications for single wall carbon nanotubes. CNI was acquired by Unidym in 2007. Dr. Gower founded Ensysce in 2008 with the focus of using single wall carbon nanotubes in therapeutic areas. Ensysce subsequently merged with Signature Therapeutics, Inc. and changed its focus to developing safe opioid pain drugs. He has served on the Board of Directors of several public companies, including Kirby Corporation, OmNova and Keystone. He also has been and continues to be involved with several not-for-profit organizations and has especially focused on Communities In Schools Houston, a leading dropout prevention and mental health program, and on Southern Illinois University with focus on the chemistry department. Dr. Gower received his B.S. from SIU and his Ph.D. from the University of Minnesota. We believe that Mr. Gower’s previous board and industry experience qualifies him to serve on our Board.
Adam S. Levin, MD joined the Board in June 2021 and is the Vice Chair of Clinical Operations for the Department of Orthopaedic Surgery at Johns Hopkins University, where he has been on faculty since 2014. He is an Associate Professor of Orthopaedic Surgery and Associate Professor of Oncology, researching treatments related to musculoskeletal oncology, while also maintaining an active clinical practice. Dr. Levin holds the Virginia M. Percy and William Algernon Percy Chair in Orthopaedic Surgery at Johns Hopkins University. He serves in additional leadership roles related to billing, coding, and practice management for the Musculoskeletal Tumor Society, the American Academy of Orthopaedic Surgeons, and the American Medical Association. Prior to joining Johns Hopkins University, Dr. Levin was an Assistant Professor of Orthopaedic Surgery at the Zucker School of Medicine at Hofstra University in New York between 2012 and 2014. From 2010 to 2012, he was a fellow of musculoskeletal oncology and Clinical Instructor at Memorial Sloan-Kettering Cancer Center, following his residency training at the North Shore/LIJ Health System (now Northwell Health) from 2005 to 2010. He has been a member of leadership fellows programs through the North Shore/LIJ Physician High Potential Program, the American Academy of Orthopaedic Surgeons, and the American Orthopaedic Association. Dr. Levin has also continued to serve as Associate Editor for CME for the Journal of Bone and Joint Surgery since 2016, and is on the Steering Committee for the Musculoskeletal Tumor Registry. Dr. Levin served as a subject-matter consultant to our predecessor, LACQ, during their initial review of our preclinical and Phase I clinical trial results. Dr. Levin holds a B.S. in Biology with a concentration in Animal Physiology from Cornell University, an M.D. from New York Medical College, and is currently studying at the Johns Hopkins University Carey School of Business for an M.B.A. with a specialization in Healthcare Management, Innovation, and Technology. We believe that Dr. Levin is qualified to serve as a member of our Board based on his academic and practice experience and his detailed knowledge of value-based care, acute and chronic pain management, novel drug design, and health care operations and management.
Steve R. Martin has served as a member of our Board since August 2020. Mr. Martin was formerly Senior Vice President and Chief Financial Officer of Armata Pharmaceuticals, Inc., a clinical development stage biotechnology company listed on New York Stock Exchange, from January 2016 until his retirement from the position on June 30, 2022. Previously, Mr. Martin served as Senior Vice President and Chief Financial Officer of Applied Proteomics, Inc., a molecular diagnostics company, from December 2014 to August 2015. From June 2011 to December 2014, Mr. Martin served as Senior Vice President and Chief Financial Officer of Apricus Biosciences, Inc. (“Apricus”), a publicly traded pharmaceutical company, and served as the Interim Chief Executive Officer of Apricus from November 2012 through March 2013. From 2008 to January 2011, Mr. Martin served as Senior Vice President and Chief Financial Officer of BakBone Software (“BakBone”), a publicly traded software company. During his final 10 months with BakBone until the company’s acquisition in January 2011, Mr. Martin also served as BakBone’s Interim Chief Executive Officer. From 2005 to 2007, Mr. Martin served as Chief Financial Officer of Stratagene Corporation, a publicly traded research products and clinical diagnostics company. Mr. Martin’s previous experience also includes serving as Controller with Gen-Probe Incorporated, a publicly traded molecular diagnostics company, as well as 10 years with Deloitte & Touche LLP, a public accounting firm. Mr. Martin holds a Bachelor in Science in Accounting from San Diego State University and is a certified public accountant (inactive). We believe that Mr. Martin’s expertise in biopharmaceutical industry and accounting expertise qualifies him to serve on our Board.
| 88 |
Lee Rauch has served on our Board since February 2022. She is an experienced Chief Executive Officer and Strategy Advisor, has served both public and private companies. During her near 40-year career, Ms. Rauch successful built companies ranging in focus from pre-clinical research to advanced clinical development, took the lead in mergers and acquisitions and used her experience to secure financing for public and private biotech companies. Among her many leadership roles, Ms. Rauch, was notably a founding member of McKinsey & Co.’s International Pharmaceutical Practice and the Executive Chairman of Springboard Enterprises Health Innovation Hub. Most recently, Ms. Rauch, served as President and CEO of Viridian Therapeutics, Inc. Ms. Rauch received a B.S. in Chemistry from Arizona State University and an M.B.A. in Finance from the University of Chicago. We believe that Ms. Rauch’s biopharmaceutical industry experience and expertise qualifies her to serve on our Board.
Curtis Rosebraugh, M.D., MPH has served on our Board since 2021. He is a member of Griebel and Rosebraugh Consulting LLC since May 2018, where he is a regulatory consultant for small molecule and biological drug development. Prior to forming a consulting firm, he was employed by the Food and Drug Administration since 2000, holding the position of Director of the Office of Drug Evaluation II (“ODEII”) within the Center for Drug Evaluation and Research (“CDER”) from 2007 until his retirement in 2018, with supervisory responsibility for the evaluation of all drug products within 3 divisions: the Division of Pulmonary, Allergy and Rheumatology Products, the Division of Metabolism and Endocrinology Products and the Division of Anesthesia, Analgesia, and Addiction Products. In this position, he has overseen the development and approval of over 50 new drugs, was responsible for the planning of over 100 advisory committee meetings, led ODE II through several controversial safety issues and has received many honors and awards. Dr. Rosebraugh has been involved in the development of abuse deterrent opioid formulations and has also been involved in the development of the biosimilar program as well as many other CDER initiatives. Dr. Rosebraugh received his undergraduate degree in pharmacy in 1981, his Medical Degree in 1986 and completed a residency in Internal Medicine in 1989, all at the University of Kansas. He completed a Masters of Public Health at Johns Hopkins School of Public Health in 1999 and a Clinical Pharmacology Fellowship at Georgetown University in 2000. We believe that Dr. Rosebraugh’s regulatory experience in the biopharmaceutical industry qualifies him to serve on our Board.
Role of Board in Risk Oversight
The Board has extensive involvement in the oversight of risk management related to us and our business and accomplishes this oversight through the regular reporting to the Board by the audit committee. The audit committee represents the Board by periodically reviewing our accounting, reporting and financial practices, including the integrity of our financial statements, the surveillance of administrative and financial controls and our compliance with legal and regulatory requirements. Through its regular meetings with management, including the finance, legal, internal audit and information technology functions, the audit committee reviews and discuss all significant areas of our business and summarize for the Board all areas of risk and the appropriate mitigating factors. In addition, our Board receives periodic detailed operating performance reviews from management.
Composition of the Board
Our business and affairs are managed under the direction of the Board. Our Board consists of eight directors, which are divided into three classes (Class I, II and III) with Class I and III each consisting of three directors and Class II consisting of two directors.
Board Committees
The standing committees of our Board consist of an audit committee, a compensation committee and a nominating and corporate governance committee. Our Board may from time to time establish other committees. Each of the committees reports to the Board.
Our president and chief executive officer and other executive officers regularly report to the non-executive directors and the audit, the compensation and the nominating and corporate governance committees to ensure effective and efficient oversight of our activities and to assist in proper risk management and the ongoing evaluation of management controls.
| 89 |
Audit Committee
We have an audit committee consisting of Steve R. Martin, who serves as the chairperson, Bob Gower and Andrew Benton. Each member of the audit committee qualifies as an independent director under the Nasdaq corporate governance standards and the independence requirements of Rule 10A-3 of the Exchange Act. Our Board has determined that Steve R. Martin qualifies as an “audit committee financial expert” as such term is defined in Item 407(d)(5) of Regulation S-K and possesses financial sophistication, as defined under the rules of Nasdaq.
The purpose of the audit committee is to prepare the audit committee report required by the SEC to be included in our proxy statement and to assist our Board in overseeing and monitoring (1) the quality and integrity of our financial statements, (2) our compliance with legal and regulatory requirements, (3) our independent registered public accounting firm’s qualifications and independence, (4) the performance of our internal audit function and (5) the performance of our independent registered public accounting firm.
Our Board adopted a written charter for the audit committee, which is available on our website.
Compensation Committee
We have a compensation committee consisting of Adam Levin, who serves as the chairperson, Bob Gower, William Chang, and Lee Rauch.
The purpose of the compensation committee is to assist our Board in discharging its responsibilities relating to (1) setting our compensation program and compensation of our executive officers and directors, (2) monitoring our incentive and equity-based compensation plans and (3) preparing the compensation committee report, if required to be included in our proxy statement under the rules and regulations of the SEC.
Our Board adopted a written charter for the compensation committee, which is available on our website.
Nominating and Corporate Governance Committee
We have a nominating and corporate governance committee, consisting of Lee Rauch, who serves as chairperson, Bob Gower, Steve R. Martin and Curtis Rosebraugh. The purpose of our nominating and corporate governance committee is to assist our Board in discharging its responsibilities relating to (1) identifying individuals qualified to become new Board members, consistent with criteria approved by the Board, (2) reviewing the qualifications of incumbent directors to determine whether to recommend them for re-election and selecting, or recommending that the Board select, the director nominees for the next annual meeting of stockholders, (3) identifying Board members qualified to fill vacancies on any Board committee and recommending that the Board appoint the identified member or members to the applicable committee, (4) reviewing and recommending to the Board corporate governance principles applicable to us, (5) overseeing the evaluation of the Board and management and (6) handling such other matters that are specifically delegated to the committee by the Board from time to time.
Our Board adopted a written charter for the nominating and corporate governance committee, which is available on our website.
Delinquent Section 16(a) Reports
None to report.
Code of Business Conduct
We adopted a code of business conduct that applies to all our directors, officers and employees, including our principal executive officer, principal financial officer and principal accounting officer, which is available on our website. Our code of business conduct is a “code of ethics,” as defined in Item 406(b) of Regulation S-K. We will make any legally required disclosures regarding amendments to, or waivers of, provisions of our code of ethics on our website.
| 90 |
Item 11. Executive & Director Compensation
This section discusses the material components of the executive compensation program for our named executive officers. Our named executive officers, consisting of our principal executive officer and the next two most highly compensated executive officers, for the year ended December 31, 2023, were:
| ● | Lynn Kirkpatrick, Ph.D., Chief Executive Officer; | |
| ● | David Humphrey, Chief Financial Officer; and | |
| ● | Geoff Birkett, Chief Commercial Officer. |
This discussion may contain forward-looking statements that are based on our current plans, considerations, expectations and determinations regarding future compensation programs. Actual compensation programs that we adopt in the future may differ materially from the currently planned programs summarized in this discussion.
Summary Compensation Table
The following table sets forth information concerning the compensation earned by our NEOs in respect of our fiscal years ended December 31, 2023 and December 31, 2022.
| Name and Principal Position | Year | Salary ($) | Bonus ($) | Stock and Option Awards ($)(1) | Non-Equity Incentive Plan Compensation ($) | All Other Compensation ($)(2) | Total ($) | |||||||||||||||||||||
| Dr. Lynn Kirkpatrick, PhD. Chief Executive Officer | 2023 | 404,875 | - | 93,800 | - | 9,900 | 508,575 | |||||||||||||||||||||
| 2022 | 392,500 | - | 214,914 | - | 15,195 | 622,609 | ||||||||||||||||||||||
Dave Humphrey Chief Financial Officer | 2023 | 338,250 | - | 93,800 | - | 9,900 | 441,950 | |||||||||||||||||||||
| 2022 | 328,333 | - | 438,562 | - | 13,722 | 780,617 | ||||||||||||||||||||||
| Geoff Birkett Chief Commercial Officer | 2023 | 312,625 | - | 70,350 | - | 9,379 | 392,354 | |||||||||||||||||||||
| 2022 | 304,167 | - | 53,664 | - | 9,125 | 366,956 | ||||||||||||||||||||||
| (1) | In accordance with SEC rules, this column reflects the aggregate grant date fair value of the restricted stock awards and stock option awards granted. This amount has been computed in accordance with Financial Accounting Standards Board (“FASB”), Accounting Standards Codification (“ASC”) Topic 718. This amount does not reflect the actual economic value that will be realized by a named executive officer upon the vesting of the stock awards or stock options, the exercise of the stock options, or the sale of the common stock underlying such awards. |
| (2) | Amounts shown represent 401(k) matching contributions. |
Narrative Disclosure to Summary Compensation Table
Elements of Compensation in 2023
The compensation of our NEOs generally consists of base salary, annual cash bonus opportunities and long-term incentive compensation in the form of equity awards, as described below.
Base Salary
The base salary payable to each NEO is intended to provide a fixed component of compensation reflecting the executive’s skill set, experience, role, responsibilities, and contributions. Base salaries were initially set at the time each NEO commenced employment with us, are reviewed annually and may be increased based on the individual performance of the NEO, company performance, any change in the executive’s position within our business, the scope of the executive’s responsibilities and any changes thereto. Effective July 1, 2023, the NEO’s annual base salary rates were $414,750 for Dr. Kirkpatrick, $346,500 for Mr. Humphrey and $320,250 for Mr. Birkett. A five percent (5%) cost of living increase was approved for executive officers, effective March 1, 2024.
| 91 |
Annual Performance-Based Bonuses
Each of our NEOs’ performance-based cash bonus opportunity is expressed as a percentage of base salary that can be achieved at a target level by meeting predetermined Company performance objectives established by the Board or the Compensation Committee. The annual bonus for Dr. Kirkpatrick is targeted at 50% of her base salary, and Mr. Humphrey and Mr. Birkett’s annual bonuses are targeted at 30% of their respective base salary. There are no amounts accrued for bonuses as of December 31, 2023.
Long-Term Equity Incentives
In 2021, the Company maintained the Ensysce Biosciences, Inc. 2021 Omnibus Incentive Plan to provide equity-based incentive awards, designed to align our interests and the interests of our stockholders with those of our employees and consultants, including our NEOs. On January 26, 2022, our stockholders approved the 2021 Amended and Restated Plan. All grants effectuated under predecessor equity plans were converted to grants outstanding under the 2021 Amended and Restated Plan. In August 2023, the 2021 Amended and Restated Plan was amended to increase the number of awards that may be granted from 31,296 to 617,092.
Employment Agreements with our NEOs
Dr. Lynn Kirkpatrick, Ph.D.
In September 2021, we entered into an employment offer letter with Dr. Kirkpatrick. The offer letter provides for Dr. Kirkpatrick’s at-will employment as our Chief Executive Officer and sets forth her annual base salary of $380,000. Additionally, the letter provides for her initial target annual bonus opportunity of up to 50% of base salary. The offer letter also indicates that Dr. Kirkpatrick is eligible to be granted certain stock awards under our equity incentive plan.
Dr. Kirkpatrick’s offer letter provides for severance benefits upon a termination of her employment by the Company without “cause”, or upon her resignation for “good reason”, in an amount equal to twelve (12) months of her then current base salary (ignoring any decrease in base salary that forms the basis for good reason); provided, however, that the payment of such benefits is subject to Dr. Kirkpatrick’s continued compliance with her obligations under her “At-Will, Confidential Information and Assignment of Inventions Agreement” and her execution of a general release of claims. Additionally, if such termination without cause or for good reason occurs within the one (1) month prior to, or during the twelve (12) month period immediately following a change in control, then all outstanding equity awards subject to time-based vesting will become fully vested on the later of Dr. Kirkpatrick’s termination date and the change in control. Cause and good reason are as defined in Dr. Kirkpatrick’s offer letter.
Dave Humphrey
In February 2021, we entered into an employment offer letter with Mr. Humphrey. The offer letter provided for Mr. Humphrey’s at-will employment as our Chief Financial Officer at an initial annual base salary of $320,000. Additionally, the letter provided for his initial target annual bonus opportunity of up to 30% of base salary. The offer letter also indicates that Mr. Humphrey is eligible to be granted certain stock awards under our equity incentive plan.
Mr. Humphrey’s offer letter provides for severance benefits upon a termination of his employment by the Company without “cause”, or upon his resignation for “good reason”, in an amount equal to six (6) months of his then current base salary (ignoring any decrease in base salary that forms the basis for good reason); provided, however, that the payment of such benefits is subject to Mr. Humphrey’s continued compliance with his obligations under his “Confidential Information and Assignment Agreement” and his execution of a general release of claims. Additionally, if such termination without cause or for good reason occurs within the one (1) month prior to, or during the twelve (12) month period immediately following a change in control, then all outstanding equity awards subject to time-based vesting will become fully vested on the later of Mr. Humphrey’s termination date and the change in control. Cause and good reason are as defined in Mr. Humphrey’s offer letter.
Geoff Birkett
In July 2021, we entered into an employment offer letter with Mr. Birkett. The offer letter provides for Mr. Birkett’s at-will employment as our Chief Commercial Officer and sets forth his annual base salary of $300,000. Additionally, the letter provides for his initial target annual bonus opportunity of up to 30% of base salary. The offer letter also indicates that Mr. Birkett is eligible to be granted certain stock awards under our equity incentive plan.
Mr. Birkett’s offer letter provides for severance benefits upon a termination of his employment by the Company without “cause”, or upon his resignation for “good reason”, in an amount equal to three (3) months of his then current base salary (ignoring any decrease in base salary that forms the basis for good reason); provided, however, that the payment of such benefits is subject to Mr. Birkett’s continued compliance with his obligations under his “Confidential Information and Assignment Agreement” and his execution of a general release of claims. Additionally, if such termination without cause or for good reason occurs within the one (1) month prior to, or the twelve (12) month period immediately following a change in control, then all outstanding equity awards subject to time-based vesting will become fully vested on the later of Mr. Birkett’s termination date and the change in control. Cause and good reason are as defined in the offer letter.
| 92 |
At-Will, Confidential Information and Assignment of Inventions Agreement
In connection with each respective offer letter, Dr. Kirkpatrick, Mr. Birkett and Mr. Humphrey entered into an “At-Will, Confidential Information and Assignment of Inventions Agreement” (the “Confidentiality Agreement”). The Confidentiality Agreements include customary prohibitions against solicitation of our customers and employees, both during employment and for two (2) years following any cessation of employment. The Confidentiality Agreements also include standard provisions relating to the Company’s intellectual property rights and prohibit the executive from disclosing confidential information. The Confidentiality Agreements are incorporated by reference into the offer letters and payment of any severance benefits under each executive’s offer letter is conditioned on continued compliance with his or her Confidentiality Agreement.
Other Benefits
We currently provide welfare benefits that are available to all of our employees, including our NEOs, including health, dental, vision and group life insurance.
Effective January 1, 2022, we put into place the Ensysce Biosciences, Inc. 401(k) Plan” (the “401(k) Plan”). The 401(k) Plan provides eligible employees with an opportunity to save for retirement on a tax-advantaged basis and under which we are permitted to make safe harbor employer contributions. Employees’ pre-tax contributions are allocated to each participant’s individual account and are then invested in selected investment alternatives according to the participants’ directions. The 401(k) Plan is intended to be qualified under Section 401(a) of the Code, with the related trust intended to be tax exempt under Section 501(a) of the Code. Under the 401(k) Plan, we make employer contributions to all employees – regardless of an employee’s contributions (or lack thereof) – in an amount equal to 3% of the employee’s eligible compensation.
We do not maintain any defined benefit pension plans or nonqualified deferred compensation plans.
Outstanding Equity Awards at Fiscal Year-End
The following table provides information regarding outstanding equity awards held by our NEOs as of December 31, 2023.
| Option Awards | Stock Awards | |||||||||||||||||||||||
| Name | Grant Date | Number of Securities Underlying Unexercised Options Exercisable (#) | Number of Securities Underlying Unexercised Options Unexercisable (#) | Option Exercise Price ($) | Option Expiration Date | Number of Shares of Stock That Have Not Vested (#) | Market Value of Shares of Stock That Have Not Vested ($) | |||||||||||||||||
| Dr. Lynn Kirkpatrick, PhD. | 1/15/2016 | 1,097 | 0 | 765.60 | 1/15/2026 | - | - | |||||||||||||||||
| 1/4/2017 | 1,920 | 0 | 439.20 | 1/4/2027 | - | - | ||||||||||||||||||
| 2/5/2018 | 3,406 | 0 | 403.20 | 2/5/2028 | - | - | ||||||||||||||||||
| 3/1/2019 | 2,743 | 0 | 621.60 | 2/28/2029 | - | - | ||||||||||||||||||
| 3/15/2019 | 27 | 0 | 621.60 | 3/14/2029 | - | - | ||||||||||||||||||
| 2/17/2022 | 381 | 452 | 336.00 | 2/17/2032 | - | - | ||||||||||||||||||
| 10/25/2023 | 100,000 | 0 | 1.18 | 10/25/2033 | - | - | ||||||||||||||||||
| Dave Humphrey | 2/4/2022 | 811 | 334 | 751.20 | 2/4/2032 | - | - | |||||||||||||||||
| 2/17/2022 | 152 | 181 | 336.00 | 2/17/2032 | - | - | ||||||||||||||||||
| 10/25/2023 | 100,000 | 0 | 1.18 | 10/25/2033 | - | - | ||||||||||||||||||
| Geoff Birkett | 10/1/2018 | 82 | 0 | 621.60 | 9/30/2028 | - | - | |||||||||||||||||
| 3/1/2019 | 1,371 | 0 | 621.60 | 2/28/2029 | - | - | ||||||||||||||||||
| 2/17/2022 | 95 | 113 | 336.00 | 2/17/2032 | - | - | ||||||||||||||||||
| 10/25/2023 | 75,000 | 0 | 1.18 | 10/25/2033 | - | - | ||||||||||||||||||
| 93 |
Pay Versus Performance
In August 2022, the SEC adopted additional disclosure requirements regarding the relationship between a registrant’s executive compensation and its financial performance. SEC rules in Item 402(v) of Regulation S-K require certain adjustments be made to the Summary Compensation Table totals to determine Compensation Actually Paid as reported in the Pay Versus Performance Table. Compensation Actually Paid does not necessarily represent cash and/or equity value earned by or paid to the applicable named executive officer without restriction, but rather is a valuation calculated under applicable SEC rules. For further information concerning our executive compensation program and our pay-for-performance philosophy, refer to the preceding compensation discussion.
The following table sets forth specified executive compensation and financial performance measures for our two most recently completed fiscal years, as required under transitional guidance for Smaller Reporting Companies provided by the SEC. We have not paid dividends and do not sponsor any pension arrangements; thus, no adjustments are made for these items.
| Year | Summary Compensation Table Total for PEO (1) | Compensation Actually Paid to PEO (2) | Average Summary Compensation Table Total for Non-PEO NEOs (3) | Average Compensation Actually Paid to Non-PEO NEOs (4) | Value of Initial Fixed $100 Investment Based on Total Shareholder Return (5) | Net Income (Loss) | ||||||||||||||||||
| 2023 | $ | 508,575 | $ | 507,187 | $ | 417,152 | $ | 416,177 | $ | 0.09 | ($ | 10,626,011 | ) | |||||||||||
| 2022 | $ | 622,609 | $ | 152,787 | $ | 573,787 | $ | 372,469 | $ | 0.80 | ($ | 25,085,496 | ) | |||||||||||
| (1) | For fiscal years 2023 and 2022, the principal executive officer (“PEO”) was our Chief Executive Officer, Dr. Lynn Kirkpatrick. | |
| (2) | The amounts disclosed reflect the following adjustments to the amounts reported in the Summary Compensation Table for the PEO: |
| Year | Less: Grant Date Value of Equity Awards | Fair Value as of Year End of Awards Granted in the Year and Outstanding and Unvested as of Year End | Change in Fair Value of Awards Granted in Prior Years and Outstanding and Unvested as of Year End | Fair Value as of Vesting Date of Awards Granted and Vested in the Year | Change in Fair Value of Awards Granted in Prior Years that Vested in the Year | Less: Fair Value as of Prior Year End of Awards Forfeited in the Year | Total Adjustments | |||||||||||||||||||||
| 2023 | $ | (93,800 | ) | $ | - | $ | (809 | ) | $ | 93,800 | $ | (579 | ) | $ | - | $ | (1,388 | ) | ||||||||||
| 2022 | $ | (214,914 | ) | $ | 1,574 | $ | - | $ | - | $ | - | $ | (256,482 | ) | $ | (469,822 | ) | |||||||||||
| (3) | For fiscal years 2023 and 2022, the Non-PEO NEOs were Dave Humphrey and Geoff Birkett. | |
| (4) | The amounts disclosed reflect the following adjustments to the amounts reported in the Summary Compensation Table for the Non-PEO NEOs: |
| Year | Less: Grant Date Value of Equity Awards | Fair Value as of Year End of Awards Granted in the Year and Outstanding and Unvested as of Year End | Change in Fair Value of Awards Granted in Prior Years and Outstanding and Unvested as of Year End | Fair Value as of Vesting Date of Awards Granted and Vested in the Year | Change in Fair Value of Awards Granted in Prior Years that Vested in the Year | Less: Fair Value as of Prior Year End of Awards Forfeited in the Year | Total Adjustments | |||||||||||||||||||||
| 2023 | $ | (82,075 | ) | $ | - | $ | (362 | ) | $ | 82,075 | $ | (613 | ) | $ | - | $ | (975 | ) | ||||||||||
| 2022 | $ | (246,113 | ) | $ | 997 | $ | - | $ | 43,798 | $ | - | $ | - | $ | (201,318 | ) | ||||||||||||
| (5) | Total Shareholder Return is calculated as the sum of (i) the cumulative amount of dividends for the measurement period, assuming reinvestment of all dividends, if any, plus (ii) the cumulative increase or decrease in the price of our common stock each respective year, divided by the price of our common stock at the beginning of the measurement period. |
| 94 |
Relationship Between Compensation Actually Paid and Company Total Shareholder Return (“TSR”)

Relationship Between Compensation Actually Paid and Net Income (Loss)
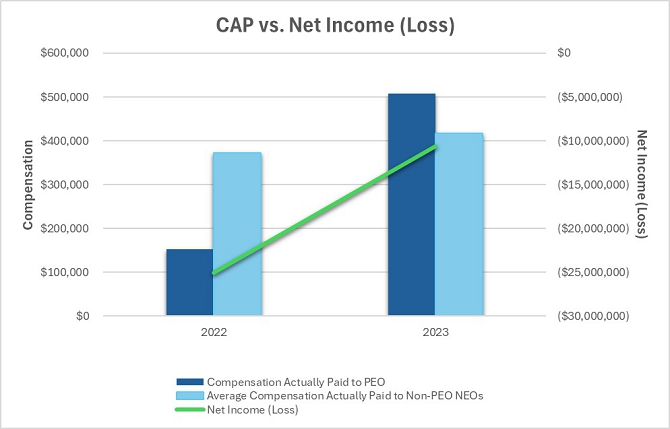
Director Compensation
The following table provides summary information concerning compensation paid or accrued by us to or on behalf of our non-employee directors for services rendered to us as of December 31, 2023.
| Name | Fees Earned or Paid in Cash ($) | Option Awards ($) | Total ($) | |||||||
| Bob Gower | 25,000 | 22,600 | 47,600 | |||||||
| William Chang | 7,500 | 22,600 | 30,100 | |||||||
| Andrew Benton | 7,500 | 22,600 | 30,100 | |||||||
| Steve Martin | 15,000 | 17,920 | 32,920 | |||||||
| Adam Levin | 10,000 | 17,920 | 27,920 | |||||||
| Lee Rauch | 11,250 | 17,920 | 29,170 | |||||||
| Curt Rosebraugh | 7,500 | 17,920 | 25,420 | |||||||
In October 2023, each Board member was granted 20,000 restricted stock units or a stock option to purchase 20,000 shares of our common stock at an exercise price of $1.13 per share. The awards were fully vested and have a ten (10) year term.
| 95 |
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The following table sets forth information with respect to the beneficial ownership of shares of our common stock by (i) each director, (ii) each named executive officer, (iii) all directors and executive officers as a group, and (iv) each person who we know beneficially owns more than 5% of our common stock as of March 8, 2024, unless otherwise indicated below.
Beneficial ownership is determined in accordance with the rules of the SEC. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities and include shares of common stock issuable upon the exercise of stock options that are immediately exercisable or exercisable within 60 days after March 8, 2024 but excludes unvested stock options. Except as otherwise indicated, all of the shares reflected in the table are shares of common stock and all persons listed below have sole voting and investment power with respect to the shares beneficially owned by them, subject to applicable community property laws. The information is not necessarily indicative of beneficial ownership for any other purpose.
In computing the number of shares of common stock beneficially owned by a person and the percentage ownership of that person, we deemed outstanding shares of common stock subject to options or warrants held by that person that are currently exercisable or exercisable within 60 days of March 8, 2024. We did not deem these shares outstanding, however, for the purpose of computing the percentage ownership of any other person.
Percentage ownership calculations for beneficial ownership for each person or entity are based on 7,329,172 shares outstanding as of March 8, 2024. Except as otherwise indicated in the table below, addresses of named beneficial owners are in care of Ensysce Biosciences, Inc., 7946 Ivanhoe Avenue, Suite 201, La Jolla, California 92037. All share amounts have been adjusted for prior reverse stock splits.
Beneficial Ownership Table
| Name and Address of Beneficial Owners | Number of Shares | Percentage | ||||||
| Officers and Directors | ||||||||
| Dr. Lynn Kirkpatrick (1) | 111,235 | 1.5 | % | |||||
| Geoff Birkett (2) | 76,565 | 1.0 | % | |||||
| David Humphrey (3) | 101,294 | 1.4 | % | |||||
| Linda Pestano (4) | 75,520 | 1.0 | % | |||||
| Andrew Benton (5) | 20,444 | * | ||||||
| William Chang (6) | 30,960 | * | ||||||
| Bob Gower (7) | 379,262 | 5.0 | % | |||||
| Adam Levin (8) | 20,145 | * | ||||||
| Steve R. Martin (9) | 20,419 | * | ||||||
| Lee Rauch (10) | 20,121 | * | ||||||
| Curtis Rosebraugh (11) | 20,145 | * | ||||||
| All directors and named executive officers as a group (eleven individuals) | 876,110 | 10.9 | % | |||||
| Greater than 5% Holders | ||||||||
| Bob Gower (7) | 379,262 | 5.0 | % | |||||
| * | Indicates less than 1%. |
| (1) | Includes 109,644 shares subject to options. |
| (2) | Consists of shares subject to options. |
| (3) | Consists of 101,086 shares subject to options. |
| (4) | Consists of shares subject to options. |
| (5) | Consists of 419 shares subject to options and 20,025 shares owned directly. |
| (6) | Includes 145 shares subject to options, 23,126 shares owned directly by Mr. Chang and his wife and 7,689 shares owned through trusts in which Mr. Chang has sole or shared voting and dispositive power. Does not include 416 shares held by trusts for family members in which Mr. Chang does not have beneficial ownership. The business address for Mr. Chang is 520 El Camino Real, 9th Floor, San Mateo, CA 94402. |
| (7) | Includes 172 shares subject to options, 121,890 shares held directly and 257,200 shares that may be acquired through the exercise of (i) warrants acquired in connection with the October 2023 Securities Purchase Agreement and (ii) warrants acquired in 2022. The business address for Mr. Gower is 101 Westcott, Unit 303, Houston, Texas 77007. |
| (8) | Consists of shares subject to options. |
| (9) | Consists of shares subject to options. |
| (10) | Consists of shares subject to options. |
| (11) | Consists of shares subject to options. |
| 96 |
Item 13. Certain Relationships and Related Transactions and Director Independence
Other than the agreements and arrangements described under the section entitled “Executive & Director Compensation” and the transactions described below, since January 1, 2023, there has not been and there is not currently proposed, any transaction or series of similar transactions to which (i) we were, or will be, a participant; (ii) the amount involved exceeded, or will exceed, $120,000 or 1% of the average of our total assets at December 31, 2022 and 2023; and (iii) in which any director, executive officer, holder of 5% or more of any class of our capital stock or any member of the immediate family of, or entities affiliated with, any of the foregoing persons, had, or will have, a direct or indirect material interest.
EBIR
We own 79.2% of the issued and outstanding shares of EBIR, a clinical stage pharmaceutical company that is developing a compound utilized in our overdose protection program for the treatment of COVID-19. The other 20.8% is owned by two affiliates of Ensysce and Mucokinetica. Specifically, our Chief Executive Officer and Director, Dr. Lynn Kirkpatrick, owns 9.9%, our former Chief Business Officer owns 9.9% and Mucokinetica owns 1.0%. Dr. Kirkpatrick is also Chief Executive Officer of EBIR. There is no revenue sharing agreement between us and EBIR.
Convertible Notes
In the 2023 Notes offering, Bob Gower, our Chairman, purchased an aggregate principal amount of Investor Notes of $216,000 for a purchase price of $200,000 and Investor Warrants that may be exercised for an aggregate of 443,187 shares of common stock. The per share conversion price of the Investor Notes and the per share exercise price of the Investor Warrants is $1.5675.
Employment Relationship
Jeff Millard is an executive officer of the Company. Mr. Millard’s spouse is also employed by the Company.
Related Party Transaction Policy
The Board previously adopted a written related person transaction policy that sets forth the following policies and procedures for the review and approval or ratification of related person transactions.
An “Immediate Family Member” means a child, stepchild, parent, stepparent, spouse, sibling, mother-in-law, father-in-law, son-in-law, daughter-in-law, brother-in-law, sister-in-law, or any person sharing the household (other than a tenant or employee).
A “Related Party” means any (a) person who is or was (since the beginning of the last fiscal year for which we have filed a Form 10-K and proxy statement, even if they do not presently serve in that role) an executive officer, director or nominee for election as a director of the Company, (b) greater than 5% beneficial owner of the Company’s outstanding common stock, or (c) Immediate Family Member of any of the foregoing.
| 97 |
A “Related Person Transaction” is any Transaction involving the Company in which a Related Party has or will have a direct or indirect material interest, as determined by the Audit Committee.
A “Transaction” means any financial transaction, arrangement or relationship or any series of similar transactions, arrangements or relationships, including indebtedness and guarantees of indebtedness and transactions involving employment and similar relationships.
Under the policy, the following types of Transactions are deemed not to create or involve a material interest on the part of the Related Party, nor will they require approval or ratification, under the policy:
| ● | Transactions involving the purchase or sale of products or services in the ordinary course of business, not exceeding $50,000 or, if the Company is a “smaller reporting company” as defined under the Securities Act, if less, one percent of the average of the Company’s total assets as of December 31st for the last two completed fiscal years. | |
| ● | Transactions in which the Related Party’s interest derives solely from his or her service as a director of another corporation or organization that is a party to the Transaction. | |
| ● | Transactions in which the Related Party’s interest derives solely from his or her ownership of less than 5% of the equity interest in another person (other than a general partnership interest) which is a party to the Transaction. | |
| ● | Transactions in which the Related Party’s interest derives solely from his or her ownership of a class of equity securities of the Company and all holders of that class of equity securities received the same benefit on a pro rata basis (e.g., dividends). | |
| ● | Transactions in which the Related Party’s interest derives solely from his or her service as a director, trustee or officer (or similar position) of a not-for-profit organization or charity that receives donations from the Company, which donations are made pursuant to the Company’s matching program, as a result of contributions by employees, that is available on the same terms to all employees of the Company. | |
| ● | Compensation arrangements of any executive officer, other than an individual who is an Immediate Family Member of a Related Party, if such arrangements have been approved or recommended to the Board for approval by the Compensation Committee. | |
| ● | Director compensation arrangements, if such arrangements have been approved by the Board or the Compensation Committee of the Board. | |
| ● | Transactions with a Related Party in which the rates or charges involved in the Transaction are determined by competitive bids, or the Transaction involves the rendering of services as a common or contract carrier, or public utility, at rates or charges fixed in conformity with law or governmental authority. | |
| ● | Indemnity payments made to directors and executive officers in accordance with the Company’s then existing certificate of incorporation, bylaws and applicable laws. | |
| ● | Transactions with a Related Party involving services as a bank depositary of funds, transfer agent, registrar, trustee under a trust indenture or similar services. |
Pursuant to its Audit Committee charter, the Audit Committee will have the responsibility to review, approve or ratify any Related Person Transactions.
| 98 |
Director Independence
Nasdaq listing rules require that a majority of the board of directors of a company listed on Nasdaq be composed of “independent directors,” which is defined generally as a person other than an officer or employee of the company or its subsidiaries or any other individual having a relationship that, in the opinion of the company’s board of directors, would interfere with the director’s exercise of independent judgment in carrying out the responsibilities of a director. Based on information provided by each director concerning his or her background, employment and affiliations, including family relationships, the Board determined that each of Bob Gower, William Chang, Andrew Benton, Steve R. Martin, Adam S. Levin, Lee Rauch and Curtis Rosebraugh is an independent director under the Nasdaq listing rules and Rule 10A-3 of the Exchange Act. In making these determinations, the Board considered the current and prior relationships that each non-employee director has and will have with us and all other facts and circumstances that the Board deems relevant in determining independence, including the beneficial ownership of our common stock by each non- employee director (and related entities) and the transactions involving them described in the section entitled “Certain Relationships and Related Party Transactions.”
Item 14. Principal Accountant Fees and Services
On April 10, 2023, the Audit Committee of our Board appointed Moss Adams LLP (“Moss Adams”) as our independent registered public accounting firm to audit our consolidated financial statements for the fiscal year ending December 31, 2023. During fiscal year 2022, Mayer Hoffman McCann P.C. (“Mayer Hoffman”) served as our independent auditor and reported on our consolidated financial statements for that year. Mayer Hoffman had been our independent auditor between 2017 and our dismissal of that firm on April 10, 2023.
The following table sets forth the aggregate fees incurred for our independent registered accounting firm for the fiscal years ended December 31, 2023 and 2022. These fees are categorized as audit fees, audit-related fees, tax fees, and all other fees. The nature of the services provided in each category is described below the table.
| 2023 | 2022 | |||||||
| Audit Fees | $ | 472,200 | $ | 670,372 | ||||
| Audit-Related Fees | 76,125 | - | ||||||
| Tax Fees | - | - | ||||||
| All Other Fees | - | - | ||||||
| Total | $ | 548,325 | $ | 670,372 | ||||
Audit fees. Consist of fees incurred for professional services rendered for the audit of the consolidated financial statements and review of the quarterly interim consolidated financial statements. These fees also include the review of registration statements and the delivery of consents in connection with registration statements. The amount for fiscal year 2023 includes $89,250 for the audit of the year ended December 31, 2022, completed in conjunction with the audit of the year ended December 31, 2023. Amounts for fiscal year 2023 reflect services performed by Moss Adams and amounts for fiscal year 2022 reflect services performed by Mayer Hoffman.
Audit-related fees. Consist of fees incurred for professional services rendered for the compliance audit in accordance with the audit requirements of Title 2 U.S. Code of Federal Regulations Part 200, Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards related to funding under federal government grants.
Tax fees. There were no fees billed for tax fees for the fiscal years ended December 31, 2023 and 2022.
All other fees. There were no fees billed for professional services rendered for other compliance purposes for the fiscal years ended December 31, 2023 and 2022.
All audit-related and other non-audit services were pre-approved by the Audit Committee, which concluded that the provision of such services was compatible with the maintenance of the respective firm’s independence in the conduct of its auditing functions.
| 99 |
PART IV
Item 15. Exhibits and Financial Statement Schedules.
Financial Statements
ENSYSCE BIOSCIENCES, INC.
CONSOLIDATED FINANCIAL STATEMENTS
| 100 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and the Board of Directors of
Ensysce Biosciences Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Ensysce Biosciences Inc. (the “Company”) as of December 31, 2023 and 2022, the related consolidated statements of operations and comprehensive income (loss), stockholders’ deficit, and cash flows for the years then ended, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the consolidated financial position of the Company as of December 31, 2023 and 2022, and the consolidated results of its operations and its cash flows for the years then ended, in conformity with accounting principles generally accepted in the United States of America.
Going Concern Uncertainty
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the financial statements, the Company has incurred recurring losses from operations and has an accumulated deficit that raise substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 2. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures to respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. We determined that there are no critical audit matters.
/s/
March 14, 2024
We have served as the Company’s auditor since 2023.
| F-1 |
Ensysce Biosciences, Inc.
Consolidated Balance Sheets
| December 31, | ||||||||
| 2023 | 2022 | |||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | $ | ||||||
| Unbilled receivable | ||||||||
| Right-of-use asset | ||||||||
| Prepaid expenses and other current assets | ||||||||
| Total current assets | ||||||||
| Property and equipment, net | ||||||||
| Other assets | ||||||||
| Total assets | $ | $ | ||||||
| Liabilities and stockholders’ deficit | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | $ | ||||||
| Accrued expenses and other liabilities | ||||||||
| Lease liability | ||||||||
| Notes payable and accrued interest | ||||||||
| Total current liabilities | ||||||||
| Long-term liabilities: | ||||||||
| Notes payable, net of current portion (at fair value) | ||||||||
| Other long-term liabilities | ||||||||
| Total long-term liabilities | ||||||||
| Total liabilities | $ | $ | ||||||
| Commitments and contingencies (Note 6) | ||||||||
| Stockholders’ deficit | ||||||||
| Preferred stock, $ par value, shares authorized, shares issued and outstanding at December 31, 2023 and December 31, 2022 | ||||||||
| Common stock, $ par value, shares authorized at December 31, 2023 and December 31, 2022; and shares issued at December 31, 2023 and December 31, 2022, respectively; and shares outstanding at December 31, 2023 and December 31, 2022, respectively | ||||||||
| Additional paid-in capital | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Ensysce Biosciences, Inc. stockholders’ deficit | ( | ) | ( | ) | ||||
| Noncontrolling interests in stockholders’ deficit | ( | ) | ( | ) | ||||
| Total stockholders’ deficit | ( | ) | ( | ) | ||||
| Total liabilities and stockholders’ deficit | $ | $ | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-2 |
Ensysce Biosciences, Inc.
Consolidated Statements of Operations
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Federal grants | $ | $ | ||||||
| Operating expenses: | ||||||||
| Research and development | ||||||||
| General and administrative | ||||||||
| Total operating expenses | ||||||||
| Loss from operations | ( | ) | ( | ) | ||||
| Other income (expense): | ||||||||
| Loss on issuance of convertible notes | ( | ) | ||||||
| Issuance costs for convertible notes | ( | ) | ||||||
| Loss on conversions and change in fair value of convertible notes | ||||||||
| Issuance of liability classified warrants | ( | ) | ||||||
| Change in fair value of liability classified warrants | ||||||||
| Interest expense, net | ( | ) | ( | ) | ||||
| Other income and expense, net | ||||||||
| Total other income (expense), net | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Net loss attributable to noncontrolling interests | ( | ) | ( | ) | ||||
| Deemed dividend related to warrants down round provision | ||||||||
| Net loss attributable to common stockholders | $ | ( | ) | $ | ( | ) | ||
| Net loss per basic and diluted share: | ||||||||
| Net loss per share attributable to common stockholders, basic and diluted | $ | ) | $ | ) | ||||
| Weighted average common shares outstanding, basic and diluted | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-3 |
Ensysce Biosciences, Inc.
Consolidated Statements of Changes in Stockholders’ Deficit
| Stockholders’ Equity (Deficit) | ||||||||||||||||||||||||
| Common Stock | Additional | |||||||||||||||||||||||
| Number of Shares | Amount | Paid-In Capital | Accumulated Deficit | Noncontrolling interests | Total | |||||||||||||||||||
| Balance on December 31, 2021 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ( | ) | |||||||||||||
| Consultant compensation | ||||||||||||||||||||||||
| Settlement of restricted stock units | ||||||||||||||||||||||||
| Conversion of convertible notes | ||||||||||||||||||||||||
| Settlement of payable to related parties | ||||||||||||||||||||||||
| Capital contribution from related parties | - | |||||||||||||||||||||||
| Public offering, net | ||||||||||||||||||||||||
| Stock-based compensation | - | |||||||||||||||||||||||
| Transaction costs associated with public offering | - | ( | ) | ( | ) | |||||||||||||||||||
| Reverse split fractional shares | ||||||||||||||||||||||||
| Deemed dividend related to warrants down round provision | - | ( | ) | |||||||||||||||||||||
| Net loss | - | ( | ) | ( | ) | ( | ) | |||||||||||||||||
| Balance on December 31, 2022 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ( | ) | |||||||||||||
| Settlement of restricted stock units | ( | ) | ||||||||||||||||||||||
| Settlement of commitment fee | ||||||||||||||||||||||||
| Conversion of convertible notes | ||||||||||||||||||||||||
| Public offering, net | ||||||||||||||||||||||||
| Stock-based compensation | - | |||||||||||||||||||||||
| Issuance of warrants - debt discount | - | |||||||||||||||||||||||
| Transaction costs associated with public offering | - | ( | ) | ( | ) | |||||||||||||||||||
| Issuance of common stock upon exercise of warrants | ( | ) | ||||||||||||||||||||||
| Reverse split fractional shares | ( | ) | ||||||||||||||||||||||
| Deemed dividend related to warrants down round provision | - | ( | ) | |||||||||||||||||||||
| Net loss | - | ( | ) | ( | ) | ( | ) | |||||||||||||||||
| Balance on December 31, 2023 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ( | ) | |||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-4 |
Ensysce Biosciences, Inc.
Consolidated Statements of Cash Flows
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Cash flows from operating activities: | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Gain on sale of asset | ( | ) | ||||||
| Accrued interest | ||||||||
| Amortization of original issue discount and debt issuance costs | ||||||||
| Loss on conversions and change in fair value of convertible notes | ( | ) | ( | ) | ||||
| Loss on issuance of convertible notes | ||||||||
| Issuance costs for convertible notes | ||||||||
| Issuance of liability classified warrants | ||||||||
| Change in fair value of liability classified warrants | ( | ) | ( | ) | ||||
| Stock-based compensation | ||||||||
| Lease cost | ( | ) | ( | ) | ||||
| Other income | ( | ) | ||||||
| Unbilled receivable | ||||||||
| Prepaid expenses and other assets | ||||||||
| Accounts payable | ( | ) | ||||||
| Accrued expenses and other liabilities | ( | ) | ||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Cash flows from investing activities: | ||||||||
| Proceeds from sale of assets | ||||||||
| Net cash provided by investing activities | ||||||||
| Cash flows from financing activities: | ||||||||
| Proceeds from public offerings, net | ||||||||
| Proceeds from issuance of convertible notes, net | ||||||||
| Repayment of convertible notes | ( | ) | ( | ) | ||||
| Transaction costs from public offerings | ( | ) | ( | ) | ||||
| Repayment of financed insurance premiums | ( | ) | ( | ) | ||||
| Net cash provided by financing activities | ||||||||
| Decrease in cash and cash equivalents | ( | ) | ( | ) | ||||
| Cash and cash equivalents beginning of period | ||||||||
| Cash and cash equivalents end of period | $ | $ | ||||||
| Supplemental cash flow information: | ||||||||
| Income tax payments | $ | $ | ||||||
| Supplemental disclosure of non-cash investing and financing activities: | ||||||||
| Stock-based compensation | $ | $ | ||||||
| Conversion of convertible notes into common stock | $ | $ | ||||||
| Payable to related parties settled in shares | $ | $ | ||||||
| Capital contribution from related parties | $ | $ | ||||||
| Original debt discount from convertible notes | $ | $ | ||||||
| Debt discount from warrants issuance | $ | $ | ||||||
| Issuance cost from convertible notes | $ | $ | ||||||
| Financed insurance premiums | $ | $ | ||||||
| Settlement of commitment fee in shares | $ | $ | ||||||
| Deemed dividend related to warrants down round provision | $ | $ | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-5 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 1 – ORGANIZATION AND PRINCIPAL ACTIVITIES
Ensysce
Biosciences, Inc. (“Ensysce”), along with its
In
2020, the Company commenced an initiative to develop a therapeutic for the treatment of certain coronavirus infections through the formation
of a separate entity, EBIR, a Delaware corporation. Pursuant to the articles of incorporation, EBIR was authorized to issue
shares of common stock, $ par value per share, and shares of preferred stock, $ par value per share. Ensysce is a
The Company currently operates in one business segment, which is pharmaceuticals. The Company is not organized by market and is managed and operated as one business. A single management team reports to the chief operating decision maker, the Chief Executive Officer.
| F-6 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 2 - BASIS OF PRESENTATION
The consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“GAAP”) and pursuant to the rules and regulations of the United States Securities Exchange Commission (“SEC”). The consolidated financial statements include the accounts of Ensysce Biosciences, Inc. and its subsidiaries. All intercompany balances and transactions have been eliminated in the consolidation.
Reverse Stock Split
In
March 2023, the Company completed a
Going Concern
The accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern, which contemplates, among other things, the realization of assets and satisfaction of liabilities in the normal course of business.
The Company has not generated any product revenue. There is no assurance that profitable operations will ever be achieved, and, if achieved, would be sustained on a continuing basis. Product development activities, clinical and pre-clinical testing, and commercialization of the Company’s product candidates are necessary to develop the Company’s products and will require significant additional financing. There can be no assurance the Company will be able to obtain such funds. These matters, among others, raise substantial doubt about the Company’s ability to continue as a going concern.
While the Company believes in the viability of its strategy to ultimately realize revenues and in its ability to raise additional funds, management cannot be certain that additional funding will be available on acceptable terms, or at all. The Company’s ability to continue as a going concern is dependent upon its ability to obtain adequate financing and achieve profitable operations. As a result, these plans do not alleviate substantial doubt about the Company’s ability to continue as a going concern for a period of 12 months following the date these consolidated financial statements were issued.
The consolidated financial statements do not include any adjustments that might be necessary should the Company be unable to continue as a going concern.
| F-7 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 3 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Use of Estimates and Assumptions
Preparation of the consolidated financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and disclosed in the accompanying notes. Actual results may differ from those estimates and such differences may be material to the consolidated financial statements. The more significant estimates and assumptions by management include, but are not limited to, the expense recognition for certain accrued research and development services.
Cash and Cash Equivalents
For purposes of the consolidated balance sheets and consolidated statements of cash flows, the Company considers all highly liquid instruments with maturity of three months or less at the time of issuance to be cash equivalents.
Concentrations of Credit Risk and Off-Balance Sheet Risk
Cash
and cash equivalents are financial instruments that are potentially subject to concentrations of credit risk. The Company’s cash
and cash equivalents are deposited in accounts at large financial institutions and amounts currently exceed federally insured limits.
The Company has no financial instruments with off-balance sheet risk of loss. Additionally, the Company had a concentration in accounts payable, as
| F-8 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
Property and Equipment
Property
and equipment include office and laboratory equipment that is recorded at cost and depreciated using the straight-line method over the
estimated useful lives of to
Fair Value Measurement
ASC 820, Fair Value Measurements, (“ASC 820”) provides guidance on the development and disclosure of fair value measurements. Under this accounting guidance, fair value is defined as an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between willing market participants at the measurement date. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or a liability.
The accounting guidance classifies fair value measurements in one of the following three categories for disclosure purposes:
| Level 1: | Quoted prices in active markets for identical assets or liabilities. | |
| Level 2: | Inputs other than Level 1 prices for similar assets or liabilities that are directly or indirectly observable in the marketplace. | |
| Level 3: | Unobservable inputs which are supported by little or no market activity and values determined using pricing models, discounted cash flow methodologies, or similar techniques, as well as instruments for which the determination of fair value requires significant judgment or estimation. |
| F-9 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
The Company evaluates assets and liabilities subject to fair value measurements on a recurring basis to determine the appropriate level at which to classify them for each reporting period. This determination requires significant judgments to be made by the Company.
As of December 31, 2023 and 2022, the recorded values of cash and cash equivalents, prepaid expenses, accounts payable, and accrued expenses and other liabilities approximate their fair values due to the short-term nature of these items.
2021 Notes
In
2021 the Company issued convertible notes with a face value of $
2022 Notes
In
July 2022 the Company issued convertible notes with a face value of $
Warrants
In 2021 the Company issued liability classified warrants in connection with the issuance of the 2021 Notes. In 2022 the Company issued liability classified warrants in connection with the issuance of the 2022 Notes. The warrants were liability classified due to certain cash settlement features and included in “Other long-term liabilities” on the consolidated balance sheets. The Company uses a Black Scholes model to estimate the fair value of the warrants. Changes in the fair value of the warrants are recognized in other income (expense) for each reporting period. Refer to Note 8 for additional details of the warrants.
The following tables present assets and liabilities measured and recorded at fair value on the Company’s consolidated balance sheet as of December 31, 2023 and 2022.
| December 31, 2023 | ||||||||||||||||
| Total | Level 1 | Level 2 | Level 3 | |||||||||||||
| Liability classified warrants | $ | $ | $ | $ | ||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||
| December 31, 2022 | ||||||||||||||||
| Total | Level 1 | Level 2 | Level 3 | |||||||||||||
| Fair value of convertible note | $ | $ | $ | $ | ||||||||||||
| Liability classified warrants | ||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||
| F-10 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
The following table summarizes the change in fair value of the Company’s Level 3 assets and liabilities for the year ended December 31, 2023:
| For the year ended December 31, 2023 | ||||||||||||
| Total | Convertible note | Liability classified warrants | ||||||||||
| Fair value, December 31, 2022 | $ | $ | $ | |||||||||
| Conversions | ( | ) | ( | ) | ||||||||
| Cash payments | ( | ) | ( | ) | ||||||||
| Cash true up liability | ( | ) | ( | ) | ||||||||
| Change in fair value | ( | ) | ( | ) | ( | ) | ||||||
| Fair value, December 31, 2023 | $ | $ | $ | |||||||||
Federal Grants
In
September 2018, the National Institutes of Health (“NIH”) through the National Institute on Drug Abuse awarded the Company
a research and development grant related to the development of its MPAR® overdose prevention technology (the “MPAR Grant”).
The total approved budget for the initial two-year period was approximately $
In
September 2019, the NIH/National Institute on Drug Abuse awarded the Company a second research and development grant related to the development
of its TAAP/MPAR® abuse deterrent technology for Opioid Use Disorder (the “OUD Grant”). The total approved budget for
the grant was approximately $
The Company recognizes revenue when costs related to the grants are incurred and assessed as reimbursable. The Company believes this policy is consistent with the overarching premise in Accounting Standards Codification Topic 606, Revenue from Contracts with Customers (“ASC 606”), applied by analogy, to ensure that it recognizes revenues to reflect the transfer of promised goods or services to customers in an amount that reflects the consideration to which it expects to be entitled in exchange for those goods or services, even though there is no “exchange” as defined in ASC 606. The Company believes the recognition of revenue as costs are incurred and reimbursable amounts become due is analogous to the concept of transfer of control of a service over time under ASC 606.
The revenue recognized under the MPAR Grant and OUD Grant was as follows:
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| MPAR | $ | $ | ||||||
| TAAP/OUD | ||||||||
| Total | $ | $ | ||||||
Amounts requested or eligible to be requested through the NIH payment management system, but for which cash has not been received, are presented as an unbilled receivable on the Company’s consolidated balance sheet. As all amounts are expected to be remitted timely, no valuation allowances are recorded.
| F-11 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
Research and Development Costs
The Company’s research and development expenses consist primarily of third-party research and development expenses, consulting expenses, animal and clinical studies, and any allocable direct overhead, including facilities and depreciation costs, as well as salaries, payroll taxes, and employee benefits for those individuals directly involved in ongoing research and development efforts. Research and development expenses are charged to expense as incurred. Payments made prior to the receipt of goods or services to be used in research and development are capitalized until the goods or services are received.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel costs associated with the Company’s executive, finance, human resources, compliance, and other administrative personnel, as well as accounting and legal professional services fees.
The Company expenses stock-based compensation over the requisite service period based on the estimated grant-date fair value of the awards using a graded amortization approach. The Company accounts for forfeitures as they occur.
The Company estimates the fair value of stock option grants using the Black-Scholes option pricing model. The assumptions used in calculating the fair value of stock-based awards represent management’s best estimates and involve inherent uncertainties and the application of management’s judgment. Stock-based compensation costs are recorded in research and development and general and administrative expenses in the consolidated statements of operations.
From time-to-time equity classified awards may be modified. On the modification date, the Company estimates the fair value of the awards immediately before and immediately after modification. The incremental increase in fair value is recognized as expense immediately to the extent the underlying equity awards are vested and on a straight-line basis over the same remaining amortization schedule as the unvested underlying equity awards.
Income Taxes
Income taxes are recorded in accordance with ASC 740, Income Taxes (“ASC 740”), which provides for deferred taxes using an asset and liability approach. The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the consolidated financial statements or tax returns. Deferred tax assets and liabilities are determined based on the difference between the consolidated financial statements and tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. Valuation allowances are provided if, based upon the weight of available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
The Company accounts for uncertain tax positions in accordance with the provisions of ASC 740. When uncertain tax positions exist, the Company recognizes the tax benefit of tax positions to the extent that the benefit would more likely than not be realized assuming examination by the taxing authority. The determination as to whether the tax benefit will more likely than not be realized is based upon the technical merits of the tax position as well as consideration of the available facts and circumstances. The Company recognizes any interest and penalties accrued related to unrecognized tax benefits as income tax expense.
| F-12 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
The basic earnings per share is calculated by dividing the Company’s net income or loss attributable to common stockholders by the weighted average number of common shares outstanding during the period. The diluted earnings per share is calculated by dividing the Company’s net earnings attributable to common stockholders by the diluted weighted average number of common shares outstanding during the period, determined using the treasury stock method and the average stock price during the period.
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Stock options | ||||||||
| RSUs | ||||||||
| Warrants | ||||||||
| Convertible Notes | ||||||||
| Total | ||||||||
Recently Adopted Accounting Pronouncements
In August 2020, the FASB issued ASU No. 2020-06, Debt – Debt with Conversion and Other Options (Topic 470) to address issues identified as a result of the complexity with applying GAAP for certain financial instruments with characteristics of liabilities and equity. The FASB decided to reduce the number of accounting models for convertible debt instruments and convertible preferred stock, resulting in fewer embedded conversion features being separately recognized from the host contract as compared with current GAAP. Certain types of convertible instruments will continue to be subject to separation models: (a) those with embedded conversion features that are not clearly and closely related to the host contract, that meet the definition of a derivative, and that do not qualify for a scope exception from derivative accounting and (b) convertible debt instruments issued with substantial premiums for which the premiums are recorded as paid-in capital. For convertible instruments, the contracts primarily affected are those with beneficial conversions or cash conversion features as the accounting models for those specific features have been removed. For contracts in an entity’s own equity, the contracts primarily affected are freestanding instruments and embedded features that are accounted for as derivatives due to a failure to meet the settlement conditions of the derivatives scope exceptions. The FASB simplified the settlement assessment by removing the requirements to (a) consider whether the contract would be settled in registered shares, (b) to consider whether collateral is required to be posted, and (c) assess shareholder rights. The FASB also decided to enhance information transparency by making targeted improvements to the disclosures for convertible instruments and earnings-per-share guidance. ASU 2020-06 is effective for fiscal years beginning after December 15, 2023 and early adoption is permitted, but no earlier than fiscal years beginning after December 15, 2020. The Company adopted the standard with an effective date of January 1, 2023 and the adoption did not have a significant impact on the consolidated financial statements.
Recently Issued Accounting Pronouncements
In November 2023, the FASB issued ASU 2023-07, “Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures”, which sets forth improvements to the current segment disclosure requirements in accordance with Topic 280 “Segment Reporting,” including clarifying that entities with a single reportable segment are subject to both new and existing segment reporting requirements. ASU 2023-07 will be effective retrospectively for fiscal years beginning after December 15, 2023 and interim periods beginning after December 15, 2024. Adoption of this ASU is currently being evaluated by the Company.
In December 2023, the FASB issued ASU No. 2023-09, “Income Taxes (Topic 740): Improvements to Income Tax Disclosures.” ASU 2023-09 requires disaggregated information about a reporting entity’s effective tax rate reconciliation as well as information on income taxes paid. ASU 2023-09 is effective for public entities with annual periods beginning after December 15, 2024, with early adoption permitted. The Company is currently evaluating the impact of this guidance on its consolidated financial statements.
| F-13 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 4 – PREPAID EXPENSES AND OTHER CURRENT ASSETS
Prepaid expenses and other current assets consisted of the following:
| December 31, | ||||||||
| 2023 | 2022 | |||||||
| Prepaid research and development | $ | $ | ||||||
| Prepaid insurance | ||||||||
| Other prepaid expenses | ||||||||
| Other current assets | ||||||||
| Total prepaid expenses and other current assets | $ | $ | ||||||
NOTE 5 – ACCRUED EXPENSES AND OTHER LIABILITIES
Accrued expenses and other liabilities consisted of the following:
| December 31, | ||||||||
| 2023 | 2022 | |||||||
| Accrued research and development | $ | $ | ||||||
| Share subscription facility commitment fees | ||||||||
| Professional fees | ||||||||
| Other accrued liabilities | ||||||||
| Total accrued expenses and other liabilities | $ | $ | ||||||
| F-14 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 6 - COMMITMENTS AND CONTINGENCIES
Purchase Commitments
As
of December 31, 2023, the Company’s commitments included an estimated $
Litigation
As of December 31, 2023 and 2022, there were no pending legal proceedings against the Company that are expected to have a material adverse effect on cash flows, financial condition or results of operations. From time to time, the Company could become involved in disputes and various litigation matters that arise in the normal course of business. These may include disputes and lawsuits related to intellectual property, licensing, contract law and employee relations matters. Periodically, the Company reviews the status of significant matters, if any exist, and assesses its potential financial exposure. If the potential loss from any claim or legal claim is considered probable and the amount can be estimated, the Company accrues a liability for the estimated loss. Legal proceedings are subject to uncertainties, and the outcomes are difficult to predict. Because of such uncertainties, accruals are based on the best information available at the time. As additional information becomes available, the Company reassesses the potential liability related to pending claims and litigation.
Lease
The
Company’s current lease agreement (as amended) has a term that extends through October 31, 2024 with no option to renew. As of
December 31, 2023, the future lease payments totaled $
Share Subscription Facility
In
December 2020, the Company executed the GEM Agreement, under which an investor agreed to provide the Company with a share subscription
facility of up to $ million for a 36-month term following the public listing of the Company’s common stock. The Company controls
the timing and maximum amount of drawdown under this facility and has no minimum drawdown obligation. The investor will pay, in cash,
a per-share amount equal to 90% of the average daily closing price of the Company’s stock during the 30 consecutive trading days
prior to the issuance of a draw notice, which shall not exceed 400% of the average trading volume for the 30 trading days immediately
preceding the draw down date. Concurrent with the public listing of the Company’s shares on July 2, 2021, the Company issued to
the investor
Compensation Subject to Shareholder Approval
In
July 2021, the Company engaged two consultants to perform certain public and investor relations services in consideration for warrants
to purchase
| F-15 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 7 - NOTES PAYABLE
The following table provides a summary of the Company’s outstanding debt as of December 31, 2023:
Principal balance | Accrued interest | Unamortized Debt Discount & Issuance Costs | Net debt balance | |||||||||||||
| 2023 Notes | $ | $ | $ | ( | ) | $ | ||||||||||
| Financed insurance | ||||||||||||||||
| Total | $ | $ | $ | ( | ) | $ | ||||||||||
The following table provides a summary of the Company’s outstanding debt as of December 31, 2022:
Principal balance | Accrued interest | Fair value adjustment | Net debt balance | |||||||||||||
| 2022 Notes | $ | $ | $ | $ | ||||||||||||
| Financed insurance | ||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||
Interest expense
The
interest expense recognized for financed insurance was $
2021 Notes
On
September 24, 2021, the Company entered into an agreement with institutional investors to issue the 2021 Notes. The agreement provided
for two closings: the first closing for $
The
2021 Notes included a stated rate of interest of
The
Company elected to apply the fair value option to the measurement of the 2021 Notes. The total initial fair value of the debt at issuance
was $
In
connection with the issuance of the 2021 Notes the Company also issued
The 2021 Notes were settled on October 11, 2022 and were not outstanding as of December 31, 2023 and 2022.
2022 Notes
On
June 30, 2022, the Company entered into an $
On
the issuance date, the Company assessed the probability of the potential settlement scenarios under the terms of the 2022 Notes and determined
that the predominant settlement feature of the 2022 Notes was the redemption feature into shares of the Company’s common stock
issuable at the lower of the conversion price or
| F-16 |
The
Company initially recorded the 2022 Notes at a fair value of $
The
December 31, 2022 fair value measurement includes the assumption of accrued interest and interest expense (at the stated rate plus an
In
connection with each of the first and second closings of the 2022 Notes, the Company also issued warrants to purchase
The
proceeds of the 2022 Notes were used for working capital purposes subject to certain customary restrictions and are secured by the Company’s
rights to its patents and licenses. The Company was restricted from issuing certain additional debt or equity without the prior written
consent of the holders for certain specified periods set forth in the 2022 Notes. If, at any time while the 2022 Notes are outstanding,
the Company carried out one or more capital raises in excess of $
The
2022 Notes were scheduled to mature on
In
January 2023, the Company entered into a letter agreement to reduce the conversion price for the remaining balance of the Company’s
outstanding 2022 Notes from $
| F-17 |
2023 Notes
On
October 23, 2023, the Company entered into a Securities Purchase Agreement (“SPA”) for an aggregate financing of $
The
combined notes are subject to an original issue discount of
The
warrants have an exercise price of $
Financed Insurance Premiums
In
June 2023, the Company renewed and financed its directors’ and officers’ liability insurance in the amount of $
NOTE 8 - STOCKHOLDERS’ EQUITY
In June 2021, in connection with the Business Combination, the Company amended and restated its Certificate of Incorporation to authorize shares of common stock and shares of preferred stock, both with par value equal to $. In September 2022, the Company amended and restated its Certificate of Incorporation to authorize up to a total of shares of common stock. As of December 31, 2023 and 2022, there were shares of preferred stock issued and outstanding.
| F-18 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
2022 December Offering
On
December 7, 2022, we entered into an underwriting agreement with an underwriter, pursuant to which we agreed to issue and sell (i)
shares of the Company’s common stock, par value $ per share, (ii) pre-funded warrants to purchase shares of common
stock and (iii) warrants to purchase
In
lieu of a purchase of common stock that would otherwise result in an investor’s beneficial ownership exceeding
Each
common warrant is exercisable immediately at an exercise price of $
2023 February Offering
On
February 2, 2023, the Company agreed to issue and sell in a registered direct offering an aggregate of shares of common stock
of the Company, par value $ per share, at an offering price of $ per share, for gross proceeds of approximately $
2023 May Offering
On
May 12, 2023, the Company completed a public offering of an aggregate of shares of its common stock at par value $ per
share (including pre-funded warrants in lieu thereof) at a combined offering price of $ per share, gross proceeds from this offering
were approximately $
In
connection with the offering, the Company also agreed to amend certain existing warrants to purchase up to an aggregate of
Warrants
On December 31, 2023, outstanding warrants to purchase shares of common stock are as follows:
| Reference | Shares Underlying Outstanding Warrants | Exercise Price | Description | Classification | ||||||||
| (a) | $ - | Equity | ||||||||||
| (b) | $ | Equity | ||||||||||
| (c) | $ | Liability | ||||||||||
| (d) | $ | Liability | ||||||||||
| (e) | $ - | Equity | ||||||||||
| (f) | $ - | Equity | ||||||||||
| (g) | $ - | Equity | ||||||||||
| (h) | $ | Equity | ||||||||||
| a) | |
| On August 3, 2021, the
Company entered into an agreement with an existing warrant holder to reduce the price of | |
| b) |
| F-19 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
| The warrants have been
subject to multiple exercise price reductions as required by a down round adjustment feature of the warrant, due to common stock
issued at prices below the then current exercise price. The adjustments have progressed from the original exercise price of $ | |
| c) | |
| d) | |
| e) |
| (f) | |
| (g) | |
| (h) |
The fair value of each warrant issued has been determined using the Black-Scholes option-pricing model. The material assumptions used in the Black-Scholes model in estimating the fair value of the warrants issued for the periods presented were as follows:
| Stock price | Exercise price | Expected term (years) | Volatility | Risk free rate | ||||||||||||||||
| (a) LACQ warrants (grant date varies) | - | % | % | |||||||||||||||||
| (b) Share subscription facility (grant date 7/2/21) | % | % | ||||||||||||||||||
| (b) Share subscription facility (remeasurement date varies) | - | - | - | % - | % | % - | % | |||||||||||||
| (c) Liability classified warrants (grant date 9/24/21) | % | % | ||||||||||||||||||
| (c) Liability classified warrants (grant date 11/5/21) | % | % | ||||||||||||||||||
| (c) Liability classified warrants (remeasured at 12/31/23) | % | % | ||||||||||||||||||
| (d) Liability classified warrants (grant date 7/1/22) | % | % | ||||||||||||||||||
| (d) Liability classified warrants (grant date 8/9/22) | % | % | ||||||||||||||||||
| (d) Liability classified warrants (remeasured at 12/31/23) | % | % | ||||||||||||||||||
In connection with the Business Combination, the Company assumed the 2021 Omnibus Incentive Plan (the “2021 Omnibus Plan”), which was approved by LACQ’s board and subsequently LACQ’s stockholders at a special stockholder meeting on June 28, 2021. The 2021 Omnibus Plan provides for the conversion with existing terms of the options outstanding under Former Ensysce stock plans and reserves for issuance an additional shares for future awards under the 2021 Omnibus Plan. No further awards may be made under the Former Ensysce stock plans.
In January, 2022, the 2021 Omnibus Plan was amended and restated to include an additional shares available for future grant and to provide for future annual increases. In February 2023, the Company’s Board of Directors approved an annual increase of shares available for future grant. In August 2023, the Company’s stockholders approved a proposal for an increase of shares available for future grant.
The Company recognized within general and administrative expense stock-based compensation expense of $ and $ for the year ended December 31, 2023 and 2022, respectively. During the year ended December 31, 2023 and 2022, the company recognized within research and development expense stock-based compensation expense of $ and $, respectively.
| F-20 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
Option Activity
During the year ended December 31, 2023, the Company granted stock options to purchase an aggregate of shares of common stock to employees and members of the board of directors. The options vested immediately and have an exercise price of between $ and $ per share. During the year ended December 31, 2022, the Company granted stock options to purchase an aggregate of shares of common stock to employees, consultants and members of the board of directors. The options vest over periods between and and have an exercise price of between $ and $ per share.
| Weighted average | ||||||||||||||||
| Options | Exercise price | Remaining contractual life | Intrinsic value | |||||||||||||
| Outstanding at December 31, 2022 | $ | $ | ||||||||||||||
| Granted | - | |||||||||||||||
| Exercised | - | |||||||||||||||
| Expired / Forfeited | ( | ) | - | |||||||||||||
| Outstanding at December 31, 2023 | ||||||||||||||||
| Exercisable at December 31, 2023 | ||||||||||||||||
| Vested and expected to vest | ||||||||||||||||
Option Valuation
| December 31, 2023 | December 31, 2022 | |||||||
| Exercise price | $ - | $ - | ||||||
| Expected stock price volatility | % - | % | % - | % | ||||
| Expected term (years) | -10.00 | |||||||
| Risk-free interest rate | % - | % | % - | % | ||||
| Expected dividend yield | % | % | ||||||
| ● | Expected stock-price volatility. The expected volatility is derived from the historical volatilities of comparable publicly traded companies within the Company’s industry over a period approximately equal to the expected term. The comparable companies were utilized as the Company’s stock does not have sufficient historical trading activity. | |
| ● | Expected term. The expected term represents the period that the stock-based awards are expected to be outstanding. The Company’s historical share option exercise experience does not provide a reasonable basis upon which to estimate an expected term due to a lack of sufficient data. Therefore, the Company estimates the expected term for employees by using the simplified method provided by the Securities and Exchange Commission. The simplified method calculates the expected term as the average of the time-to-vesting and the contractual life of the options. | |
| ● | Risk-free interest rate. The risk-free interest rate is based on the U.S. Treasury yield in effect at the time of grant for zero coupon U.S. Treasury notes with maturities approximately equal to the expected term. | |
| ● | Expected dividend yield. The expected dividend is assumed to be zero as the Company has never paid dividends and has no current plans to pay any dividends on the Company’s common stock. |
| F-21 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
The weighted-average grant date fair value of options granted during the year ended December 31, 2023 was $. The weighted-average grant date fair value of options granted during the year ended December 31, 2022 was $.
As of December 31, 2023, the Company had an aggregate of $ of unrecognized share-based compensation cost, which is expected to be recognized over the weighted average period of years.
Restricted Stock Units
| Restricted Stock Units | Weighted average fair value | |||||||
| Outstanding at December 31, 2022 | $ | |||||||
| Granted | ||||||||
| Released | ( | ) | ||||||
| Cancelled | ( | ) | ||||||
| Outstanding at December 31, 2023 | $ | |||||||
The estimated fair value of each of the Company’s restricted stock unit awards granted in 2023 was determined on the date of grant based on the closing price of the Company’s common stock on the previous trading date. The restricted stock unit awards granted in 2023 were immediately vested and there were no other valuation inputs used for the estimated fair value.
Shares Reserved for Future Issuance
The following shares of common stock are reserved for future issuance:
| December 31, 2023 | ||||
| Awards outstanding under the 2021 Omnibus Incentive Plan | ||||
| Awards available for future grant under 2021 Omnibus Incentive Plan | ||||
| Warrants outstanding | ||||
| Total shares of common stock reserved for future issuance | ||||
NOTE 10 - INCOME TAXES
Loss before provision for income taxes consisted of the following:
| Year ending December 31, | ||||||||
| 2023 | 2022 | |||||||
| United States | $ | ( | ) | $ | ( | ) | ||
The federal and state income tax provision (benefit), included in general and administrative expenses in the Consolidated Statement of Operations, is summarized as follows:
| Year ending December 31, | ||||||||
| 2023 | 2022 | |||||||
| Current state provision | $ | $ | ||||||
| F-22 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
The effective tax rate of the Company’s provision (benefit) for income taxes differs from the federal statutory rate as follows:
| Year ending December 31, | ||||||||
| 2023 | 2022 | |||||||
| Income (benefit) taxes at statutory rates | ( | ) | ( | ) | ||||
| State income tax, net of federal benefit | ( | ) | ( | ) | ||||
| Warrants and convertible debt | ( | ) | ( | ) | ||||
| Nondeductible executive compensation | ||||||||
| Stock based compensation | ||||||||
| Share subscription facility transaction costs | ||||||||
| Research and development tax credits | ( | ) | ( | ) | ||||
| Change in tax rates | ( | ) | ||||||
| Other | ( | ) | ||||||
| Change in valuation allowance | ||||||||
| Total | ||||||||
Deferred income taxes reflect the net tax effects of (a) temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes, and (b) operating losses and tax credit carryforwards.
The Company’s deferred tax assets were comprised of the following:
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss tax carryforwards | $ | $ | ||||||
| Tax credits | ||||||||
| Capitalized research costs | ||||||||
| Stock-based compensation | ||||||||
| Other | ||||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Total deferred tax assets | ||||||||
| Deferred tax liabilities: | ||||||||
| Other | ( | ) | ||||||
| Total deferred tax liabilities | ( | ) | ||||||
| Net deferred tax assets | $ | $ | ||||||
As
of December 31, 2023, the Company had federal and California net operating loss (NOL) carryforwards of $
| F-23 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
In
addition, as of December 31, 2023, the Company had federal and state research and development (R&D) tax credit carryforwards of $
Pursuant
to IRC Sections 382 and 383, annual use of the Company’s NOL and R&D credit carryforwards may be limited in the event that
a cumulative change in ownership of more than 50% occurs within a three-year period. Although the Company has not completed an IRC Section
382/383 analysis regarding the limitation of NOL and R&D credit carryforwards as of December 31, 2023, the Company estimates that
approximately $
The following table summarizes the activity related to the Company’s unrecognized tax benefits:
| Year ending December 31, | ||||||||
| 2023 | 2022 | |||||||
| Balance at beginning of year | $ | $ | ||||||
| Increases related to current year tax positions | ||||||||
| Increases related to prior year tax positions | ||||||||
| Decreases related to prior year tax positions | ||||||||
| Balance at end of year | $ | $ | ||||||
As
of December 31, 2023 and 2022, the Company had unrecognized tax benefits of $
The Company and its subsidiaries are subject to U.S. federal income tax as well as income tax in multiple state jurisdictions. With few exceptions, the Company is no longer subject to United States federal income tax examinations for years before 2020 and state and local income tax examinations before 2019. However, to the extent allowed by law, the tax authorities may have the right to examine prior periods where net operating losses were generated and carried forward, and make adjustments up to the amount of the NOL carryforward amount. The Company is not currently under examination by the Internal Revenue Service or any state or local tax authority.
| F-24 |
Ensysce Biosciences, Inc.
Notes to the Consolidated Financial Statements
NOTE 11 - RELATED PARTIES
In
July 2022, the Chief Executive Officer and a Board member transferred shares of registered common stock to GYBL to settle $
In
connection with the issuance of the 2023 Notes, the Company issued to a board member a $
NOTE 12 - SUBSEQUENT EVENTS
In
January and February 2024, the Company issued million shares of common stock in repayment of $
In
February 2024, the Company entered into definitive agreements for the immediate exercise of certain outstanding warrants to purchase
up to an aggregate of
| F-25 |
Exhibit Index
| 101 |
| 102 |
| * | Filed herewith. |
| † | Certain schedules (or similar attachments) to this Exhibit have been omitted in accordance with Regulation S-K Item 601(a)(5) or 601(b)(2), as applicable. The registrant agrees to furnish supplementally a copy of all omitted schedules to the Securities and Exchange Commission upon its request. |
| + | Denotes compensatory plans or arrangements or management contracts. |
| ++ | This certificate accompanies this report pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 and shall not be deemed filed by Ensysce for purposes of Section 18 or any other provisions of the Exchange Act. |
Item 16. Form 10-K Summary.
Not applicable.
| 103 |
SIGNATURES
Pursuant to the requirements of the Securities Act, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized, in San Diego, State of California, on March 14, 2024.
| ENSYSCE BIOSCIENCES, INC. | ||
| By: | /s/ Dr. Lynn Kirkpatrick | |
| Name: | Dr. Lynn Kirkpatrick | |
| Title: | President, Chief Executive Officer and Director | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed by the following persons in the capacities indicated on March 14, 2024.
| Name | Title | ||
| By: | /s/ Dr. Lynn Kirkpatrick | President, Chief Executive Officer and Director | |
| Dr. Lynn Kirkpatrick | (Principal Executive Officer) | ||
| By: | /s/ David Humphrey | Chief Financial Officer, Secretary and Treasurer | |
| David Humphrey | (Principal Financial and Accounting Officer) | ||
| By: | /s/ Andrew Benton | Director | |
| Andrew Benton | |||
| By: | /s/ William Chang | Director | |
| William Chang | |||
| By: | /s/ Bob Gower | Director and Chairman of the Board | |
| Bob Gower | |||
| By: | /s/ Adam Levin | Director | |
| Adam Levin | |||
| By: | /s/ Steve Martin | Director | |
| Steve Martin | |||
| By: | /s/ Lee Rauch | Director | |
| Lee Rauch | |||
| By: | /s/ Curtis Rosebraugh | Director | |
| Curtis Rosebraugh | |||
| 104 |